Does Anti-Amyloid Immunotherapy Need the Lymphatic System?
Quick Links
Why does anti-amyloid immunotherapy clear plaque faster in some people than others? In the April 28 Nature, researchers led by Jonathan Kipnis, Oscar Harari, and Carlos Cruchaga at Washington University in St. Louis suggest that the health of the meningeal lymphatic system may be a factor. This system consists of tiny vessels that pervade the brain’s outer covering and help drain fluid and solutes from parenchymal tissue. When Kipnis and colleagues destroyed some of these vessels in a mouse model of amyloidosis, anti-amyloid antibodies cleared less plaque from the brain. Conversely, when the scientists stimulated growth of new lymphatic vessels, antibodies mopped up plaque more efficiently.
- In mice, damaging meningeal lymphatic vessels slows plaque clearance by aducanemab and lecanemab.
- Enhancing lymphatic vessels boosts plaque clearance.
- Dysfunctional lymphatics activate microglia.
The condition of lymphatic vessels on the brain’s surface affected the mice’s vasculature and immune systems, as well. After vessel ablation, endothelial cells in blood vessels expressed more vascular repair genes, while microglia assumed a disease-associated microglia (DAM) state. This microglial profile resembled that of activated microglia from postmortem sections of Alzheimer’s brain, the authors found.
Helene Benveniste at Yale School of Medicine in New Haven, Connecticut, was particularly impressed by the potential causal link between impaired lymphatics and microglial activation. “This is a breakthrough study,” she told Alzforum. Mony de Leon at Weill Cornell Medicine College in New York City was likewise intrigued, though he noted there are still many mechanistic questions about how these systems interact. “This paper is a good first step that generates a lot of hypotheses,” he told Alzforum.
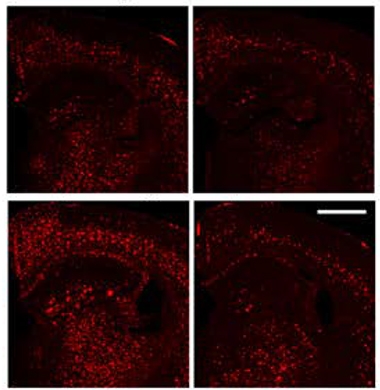
Stuck Plaques. In 5XFAD mice (top left), anti-amyloid antibodies (top right) clear plaque (red). When lymphatic vessels are damaged, the mice accumulate more plaque (bottom left), and antibodies clear it poorly (bottom right). [Courtesy of Da Mesquita et al., Nature.]
The study of brain lymphatics is quite new. Researchers including Kipnis first identified meningeal vessels in 2015 (Oct 2017 news). These tiny tubes are similar to blood vessels, but are located outside the brain in the dura mater. Instead of blood, they carry cerebrospinal fluid drained from the brain.
Since their discovery, studies have called this system the main exit route for CSF, with most of the fluid flowing through basal lymphatic vessels to lymph nodes in the neck, and from there to blood (Nov 2017 news; Jul 2019 news). Kipnis and colleagues previously showed that drainage efficiency in the mouse brain wanes with age. This may be bad for the brain, since damaged lymphatic vessels weakened memory in young, wild-type mice, and exacerbated plaque buildup in a mouse model of amyloidosis (Jul 2018 news).
In the current study, the authors investigated how the lymphatic system might affect the efficacy of anti-amyloid antibodies. Scientists still do not fully understand how these antibodies clear Aβ from the brain. First author Sandro Da Mesquita injected the light-activated toxin verteporfin into the CSF of young 5XFAD mice, then shone light through the skull onto the dorsal lymphatic vessels. This damaged those vessels. The researchers then treated these animals, and intact controls, with eight weekly doses of mouse analogs of aducanumab or lecanemab. Compared to 5XFAD mice with intact lymphatics, the damaged animals cleared less plaque (see image above). They also had more dystrophic neurites and learned more slowly in the Morris water maze.
What caused these problems? In the damaged mice, about 20 percent fewer antibodies reached plaques in the first place, but Kipnis believes the main reason plaque persisted was the slower removal of material from the brain. Clogging clearance pathways had wide-reaching effects. Endothelial cells in blood vessels turned on genes involved in vascular activation, cell migration, and proliferation. Perhaps impairing lymphatic drainage puts more pressure on the capillaries of the blood-brain barrier to clear waste, leading to these changes, Kipnis suggested.
Meanwhile, mice with damaged lymphatic vessels had about 50 percent more activated microglia than controls, and these cells expressed a pro-inflammatory, DAM-like signature (Jun 2017 news). Kipnis speculated that when the antibodies busted up plaque, they freed toxic Aβ oligomers that lingered in the brain and revved up the microglia.

Vessel Expansion. After treatment with VEGF-C (bottom), lymphatic vessels (green) expand compared to control mice (top). Skull is blue. [Courtesy of Da Mesquita et al., Nature.]
Enhancing lymphatic vessels, on the other hand, boosted clearance. Da Mesquita and colleagues expressed vascular endothelial growth factor C (VEGF-C) in 5-month-old 5XFAD mice. This growth factor preferentially binds a receptor on lymphatic endothelial cells, but can also affect the vascular endothelium (Joukov et al., 1996). As expected, treated mice grew more lymphatic vessels, and in these animals, lecanemab cleared about twice as much plaque as in control 5XFADs. They did not try the aducanumab analog in this experiment.
The benefit of enhancing the lymphatic system was greater in aged mice. In 2-year-old APPswe and J20 mice, treatment with aducanumab alone failed to budge plaque. When combined with VEGF-C, however, up to half of plaques were wiped out.
Researchers were enthusiastic about these data. “This is a proof of principle that immunotherapy works best if you have an efficient lymphatic system,” Costantino Iadecola, also at Weill Cornell Medical in New York, told Alzforum. Matthias Nahrendorf at Massachusetts General Hospital, Boston, noted that the findings might apply to other therapies directed to the brain, as well. “It may be highly clinically relevant,” he wrote (full comment below).
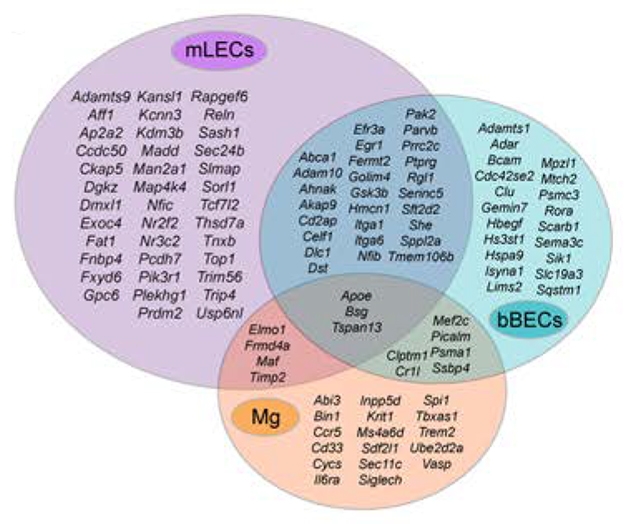
Overlooked in AD? Mouse lymphatic endothelial cells (purple) and vascular brain endothelial cells (aqua) express more AD-related genes than do microglia (orange), hinting at a role for lymphatic processes in disease. [Courtesy of Da Mesquita et al., Nature.]
Do the findings apply to people? To get at this, Da Mesquita and colleagues compared their mouse expression data with known AD genes. Surprisingly, mouse lymphatic endothelial cells expressed a greater number of those human risk genes than did cortical microglia. The set included such stalwarts as APOE, SORL1, and ABCA1 (see image above). In cohort studies at WashU, inheritance of AD risk SNPs from this lymphatic gene set correlated with higher amyloid load, higher AD risk, and a lower age of onset. Likewise, in human postmortem brain samples, the same SNPs correlated with having a larger number of activated microglia that expressed AD-associated genes. These activated microglia resembled those seen in mice with damaged lymphatic systems. Altogether, Kipnis believes the findings imply that lymphatic dysfunction could contribute to AD pathogenesis.
Greg Lemke at the Salk Institute for Biological Studies, La Jolla, California, agreed. “This strengthens the connection between meningeal lymphatic impairment and aberrant microglial activation in AD, though the mechanism requires further study,” he wrote to Alzforum (see comment below).
To explore the mechanism, Kipnis’ group will analyze lymphatic endothelial cell gene expression in 24 AD brains and six healthy, age-matched controls. He wants to determine if there is a consistent disease signature. Because endothelial cells are fragile and hard to isolate, the scientists will use fresh autopsy tissue. Kipnis also plans to investigate what AD-linked genes do in lymphatic endothelial cells.
Kipnis is also interested in the study’s implications for immunotherapy. If the health of the lymphatic system determines how well antibodies work, and this is mediated by genetics, then it might be possible to define a genetic signature to stratify patients into those who will respond best, he suggested.
Kipnis has partnered with Boston-based biotech company PureTech Health to search for therapeutic strategies to boost lymphatic drainage. One option is to find a method for delivering VEGF-C to people in a targeted way so that it only affects lymphatic vessels. Because the meningeal lymphatic system lies outside the central nervous system, it is easier to target than brain tissue, Kipnis noted.
Other researchers cautioned that the brain’s drainage system is complex, integrating perivascular clearance, glymphatic flow, and transcytosis through the blood-brain barrier. Many parts of this system break down with aging, so improving lymphatic vessels may not fix everything, Iadecola noted. De Leon believes more work is needed to understand how the kinetics and mechanisms of clearance vary between different brain regions. He suggested studying how impaired lymphatics might affect CSF biomarkers of AD.—Madolyn Bowman Rogers
References
News Citations
- Lymphatic Vessels Found in Human Brain
- In Mice, CSF Caught Draining Via Lymphatic Vessels, Not Veins
- Anatomy News Flash: Brain Drains Lymphatic Fluid Through Its Base
- Lymphatic Brain Drain Withers in Aging, Worsens Disease
- Hot DAM: Specific Microglia Engulf Plaques
Research Models Citations
Paper Citations
- Joukov V, Pajusola K, Kaipainen A, Chilov D, Lahtinen I, Kukk E, Saksela O, Kalkkinen N, Alitalo K. A novel vascular endothelial growth factor, VEGF-C, is a ligand for the Flt4 (VEGFR-3) and KDR (VEGFR-2) receptor tyrosine kinases. EMBO J. 1996 Jan 15;15(2):290-98. PubMed.
Further Reading
News
- Ring Around the Vessel: Enlarged Spaces Signal Vascular Disease
- Mini Strokes Cause Mega Problems for Brain Cleansing
- Amyloid Plaques’ Hold on Aβ42 Dampens Peptide’s Daily Rhythm
- Dearth of Water Channels a Sign of ‘Glymphatic’ Breakdown in Alzheimer’s?
- LOAD of Data Place Vascular Malfunction as Earliest Event in Alzheimer’s
- Sleep and Brain Cleansing—Fresh Insights into Regulation and Disruption
- Brain Drain—“Glymphatic” Pathway Clears Aβ, Requires Water Channel
Primary Papers
- Da Mesquita S, Papadopoulos Z, Dykstra T, Brase L, Farias FG, Wall M, Jiang H, Kodira CD, de Lima KA, Herz J, Louveau A, Goldman DH, Salvador AF, Onengut-Gumuscu S, Farber E, Dabhi N, Kennedy T, Milam MG, Baker W, Smirnov I, Rich SS, Dominantly Inherited Alzheimer Network, Benitez BA, Karch CM, Perrin RJ, Farlow M, Chhatwal JP, Holtzman DM, Cruchaga C, Harari O, Kipnis J. Meningeal lymphatics affect microglia responses and anti-Aβ immunotherapy. Nature. 2021 May;593(7858):255-260. Epub 2021 Apr 28 PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
Salk Institute
Salk Institute
This paper adds to the important literature from the Kipnis lab that establishes the brain’s meningeal lymphatics as a critical interface between the nervous and immune systems. Using photoablation protocols to eliminate the lymphatic vasculature of the dorsal meninges in mice, Da Mesquita and colleagues demonstrate that this vasculature is important for the ability of anti-Aβ antibodies to reduce plaque burden in amyloidogenic mouse models of Alzheimer’s disease (AD). Conversely, the authors show that enhancing lymphatic vascular integrity through expression of vascular endothelial growth factor C potentiates the Aβ plaque-clearing efficacy of these antibodies, human versions of which are currently in clinical trials or awaiting approval as AD therapeutics.
Using transcriptomic profiling, the authors identify genetic similarities between microglia from 5xFAD brains with ablated meningeal lymphatics and human microglia from AD patients, suggesting a potential role of compromised brain lymphatics in human AD. They also note that AD genetic variants that are expressed by lymphatic endothelial cells are associated with altered microglial gene expression in human cortex. This strengthens the connection between meningeal lymphatic impairment and aberrant microglial activation in AD, though the mechanism behind this connection requires further study.
CNS lymphatics have been overlooked in health and disease until recently, and there is little known about how dysfunction in meningeal lymphatic drainage might affect the outcome of immunotherapy in AD. This study is therefore topical, as it provides an alternative perspective with respect to results from current and past immunotherapies for AD.
I think this is very interesting work, and the authors deserve credit for looking at parallels between human disease and the mouse models used. Clinically, the work may be highly relevant since the data imply that meningeal lymphatics are important for drug delivery. This makes a lot of sense to me, as this type of drug delivery problem is a frequent issue in many settings, for instance in cancer. It is likely even more relevant because of the insulating nature of the blood-brain barrier.
Overall, the meninges and brain lymphatics are increasingly getting attention, and I believe rightly so. They provide important routes (or barriers) to the brain, especially for wandering cells, such as immune cells.
University of Southampton School of Medicine
This paper is very valuable in connecting the neuroimmunology of the meningeal lymphaticswith microglia—the innate immune system for the brain, especially in the context of immunization against Aβ.
It is interesting that in mice with impaired meningeal lymphatic drainage the administration of the mAb158 antibody resulted in less mAb158 co-localizing with the parenchymal plaques, but no effect on the amount of mAb158 co-localizing with vascular Aβ. This suggests that meningeal lymphatic biology is directly connected to the innate immune system of the brain, but more investigations into its connections with the intramural periarterial drainage pathways for the clearance of interstitial fluid are required.
BioArctic
Uppsala University
In this interesting paper, the authors demonstrate the importance of an intact meningeal lymphatic system for successful passive Aβ immunotherapy. Furthermore, gene signature data, from both a mouse AD model and AD patients, suggest a link between meningeal lymphatic dysfunction and microglia activation.
The authors provide convincing data that therapeutic antibodies directed toward Aβ are less efficient in entering brain parenchyma and consequently, they largely fail to clear Aβ plaques following induced disruption of meningeal lymphatic vessels. They demonstrate that by restoring lymphatic vessel integrity by viral VEGF-C expression, antibody-mediated Aβ clearance was improved.
As there was significantly less co-localization of the administrated anti-Aβ antibody with brain parenchymal Aβ aggregates, it is likely that the circulation between cerebral spinal fluid, glymphatic flow, and interstitial fluid has been compromised by the loss of lymphatic vasculature, thereby lowering the brain exposure through this specific route. A more direct pathway into the brain is to cross the blood-brain barrier (BBB). Different technologies are now being developed to transport biotherapeutics over the BBB so it would be interesting to investigate if a BBB transport approach is affected by meningeal lymphatic dysfunction. This would help to understand if the lower clearance of Aβ plaques with an anti-Aβ antibody in mice with loss of lymphatic vasculature is due to less drainage capacity or a direct reduction in brain exposure of the antibody.
The development of a gene signature of early meningeal lymphatic dysfunction, potentially aligned with microglial activation and angiopathy, has the potential to allow patient stratification for immunotherapies of AD. Enhancement of meningeal lymphatic function combined with immunotherapy might in the future lead to better clinical outcomes.
University of Eastern Finland
This interesting paper convincingly shows that elimination of meningeal lymphatics by photoablation increases the amyloid plaque load in the cortices and thalami of 5xFAD mice. However, the claim that “Compromised meningeal lymphatic function in 5xFAD mice limits brain Aβ clearance by chimeric mAducanumab” (Fig. 1 heading) needs to be interpreted with caution. The claim is based on Fig. 1d showing significantly larger amyloid plaque coverage per brain section in mAb-treated mice with photoablation than in treated mice with intact menigeal lymphatics in a paired comparison.
This is only a partial view of the big picture. The study had a typical 2 x 2 design and should be analyzed with two-way ANOVA. This would have shown a significant main effect of the photoablation, as well as a significant effect of the mAb treatment. However, there is no interaction between these two factors, which would support the given claim.
In fact, Fig. 1c, presenting the amyloid plaque number per mm2 with all four study groups, shows a similar a mAb treatment effect in mice with and without photoablation. Fig. 1d would most likely look the same had all the four groups been shown.
Further, the analysis of dystrophic neurites stained by LAMP1 (Fig 1e) might actually show a significant lesion by mAb treatment interaction but to the opposite direction as claimed. The treatment effect is minimal in mice with intact meningeal lymphatics (due to overall small number of these dystrophies) but highly significant in mice with ablated lymphatics. Thus, the correct interpretation of the data is that photoablation of meningeal lymphatics results in increased brain amyloid load but does not mitigate the Aβ clearance by systemic administration of mAducanumab.
Interestingly, the situation is somewhat different when the antibody (this time mAb158) is administered directly to CSF. As seen in Extended Fig. 5e and g, the number of plaques per mm2 and plaque coverage per brain section are reduced by the mAb treatment in mice with intact meningeal lymphatics, but not in mice with photoablation. It is a pity that this second study was carried out with a different mAb than the first one, leaving open the question whether the difference is due to the route of administration or the antibody itself. The different outcomes of meningeal lymphatic ablation on systemic versus local anti-Aβ mAb warrant further studies.
Make a Comment
To make a comment you must login or register.