Anatomy News Flash: Brain Drains Lymphatic Fluid Through Its Base
Quick Links
The 2015 discovery of lymphatic vessels in the brain’s meninges threw open a door to seeing how this still-mysterious organ clears waste. Subsequent work showed that these vessels, which reside in the brain’s outermost membrane, the dura mater, help drain large molecules and immune cells from cerebrospinal fluid to the periphery. It remained unclear, however, how molecules pass from inside the brain into these meningeal vessels. Now, in the July 24 Nature online, researchers led by Sung-Hong Park and Gou Young Koh at the Korea Advanced Institute of Science and Technology in Daejeon, South Korea, make the case that this occurs in vessels located at the base of the brain. In mice, they found that these vessels have the right anatomical features and are well-positioned spatially to take up solutes from CSF. Functional studies appeared to confirm this when labeled tracers were seen leaving the brain via this route.
- Basal, but not dorsal, lymphatic vessels specialize in fluid uptake.
- Tracers travel through basal lymphatic vessels to reach cervical lymph nodes.
- Those vessels become misshapen with age, slowing transport.
Notably, transport was less efficient in old mice, in whom those basal lymphatic vessels were enlarged and misshapen. Clearance of brain proteins is known to slow with age.
“The identification of the precise exit routes for fluids leaving the brain is a crucial step toward understanding how waste is cleared from the CNS,” Taija Mäkinen at Uppsala University, Sweden, wrote in an accompanying Nature editorial. “This finding might eventually enable the development of therapies that promote CNS drainage to combat pathological processes in neurological diseases.”
Others agree this is a step forward. “The study combines creative noninvasive MRI and microscopy at a technically very high level, and adds to our understanding of how lymph is drained from the CNS,” Matthias Nahrendorf at Massachusetts General Hospital, Boston, wrote to Alzforum. He also cautioned that it remains unknown whether the same route functions in humans.
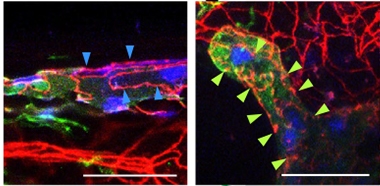
Zippers, Buttons. In dorsal lymphatic vessels (left), endothelial cells (green; nuclei blue) adhere tightly to each other as though zipped (continuous red lines), while in basal vessels (right), cells adhere in discrete spots, like buttons (broken red lines), allowing fluid to pass. [Courtesy of Ahn et al., Nature.]
Although putative lymphatic vessels in the brain were first identified by the 17th-century anatomist Paolo Mascagni, 20th-century scientists doubted their existence. In 2015, two separate groups unambiguously identified lymphatic vessels in mouse dura and showed that they drained solutes from CSF. Human verification followed (Nov 2017 news; Oct 2017 news). How solutes enter these vessels remained a puzzle, however. The vessels were most apparent on the top of the brain, where they were separated from brain fluid by a thick dural layer. A recent study found that tracer traveling from brain to lymph nodes did not run through dorsal lymphatic vessels (Ma et al., 2017).

Special Architecture. Lymphatic vessels at the top of the brain (left) have tightly sealed junctions, while those at the base (right) have branched endings where endothelial cells are loosely buttoned together. [Courtesy of Ahn et al., Nature.]
Park and Koh wondered if lymphatic vessels elsewhere in the brain might handle drainage instead. Vessels at the base of the skull have been hard to study because of the complicated bony architecture there. To visualize these structures, first author Ji Hoon Ahn used Prox1-GFP mice, which express a fluorescent reporter in lymphatic endothelial cells (Choi et al., 2011). Examining basal vessels in these mice, the scientists found that they were highly branched, with numerous rounded ends, and possessed features characteristic of capillaries.
Individual endothelial cells in the vessel walls had a multilobed shape, like oak leaves. Instead of being tightly sealed together along their whole length, as a zipper is, the cells were pegged together at discrete spots as though buttoned (see image above). These “button-like” junctions leave gaps where fluid can enter. The vessels contained internal valves that help prevent fluid from running backward. In contrast, dorsal lymphatic vessels have zipper-like junctions and no valves. Moreover, basal lymphatic vessels lacked a covering sheath of smooth muscle cells. These features would allow the basal vessels to absorb and transport CSF, the scientists reasoned.

Easy Access. In dorsal mouse brain (left), lymphatic vessels (green) are separated from brain by a thick fibrous layer (blue) and strong arachnoid barrier (red), while in basal brain (right), vessels lie close to a flimsy arachnoid layer. [Courtesy of Ahn et al., Nature.]
The surroundings of the basal lymphatic vessels suggested they were in the right location to interact with CSF. They course through a thin dural layer that measures only one or two cells across. They lie close to the subarachnoid space, separated from it by only a loose, mesh-like arachnoid barrier. Dorsal lymphatic vessels, on the other hand, lie in a thick, fibrous dural layer and are separated from CSF by a tight arachnoid membrane (see image at right).

Basal Clearance. Tracer (red arrows) enters branches (green) of lymphatic vessels at the back of the brain. [Courtesy of Ahn et al., Nature.]
To test whether basal lymphatic vessels indeed drain the brain, the authors infused a large-molecule tracer, Quantum Dot 705, into the brain parenchyma or subarachnoid space of Prox1-GFP mice. In either case, the signal appeared in basal lymphatic vessels within five minutes, then exited the skull and accumulated in cervical lymph nodes. The signal never appeared in dorsal lymphatic vessels. In other experiments, the authors observed T cells passing directly from brain into basal lymph vessels, suggesting immune cells might leave the brain via this route, as well.
What happens with age? Two-year-old mice had fewer valves in their basal lymphatic vessels, and those that remained were misshapen. Those vessels were larger and more branched, possibly to compensate for poor drainage, the authors suggested. Supporting this, the aged vessels poorly cleared Quantum Dot 705 from brain (see image below right). Given that clearance of pathological protein aggregates such as Aβ42 slows with age, this finding may inform what goes awry in neurodegenerative diseases, the authors believe.
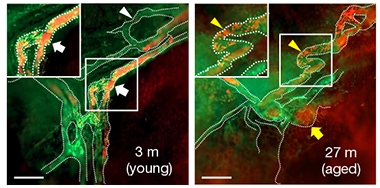
Slowdown With Age. In young mice (left), a large-molecule tracer (red) drains quickly through basal lymphatic vessels (green); in 2-year-old mice (right), tracer moves sluggishly through enlarged, twisted vessels. [Courtesy of Ahn et al., Nature.]
The findings in aged mice replicate previous work from Jonathan Kipnis and colleagues at the University of Virginia, Charlottesville, who saw CSF drainage from brain to lymph nodes slow down in aged wild-type mice. In that study, ablating meningeal vessels in 5xFAD mice led to more amyloid plaque buildup in the hippocampus and meninges, suggesting this disposal route is crucial for controlling Aβ levels (Jul 2018 news). “Step by step, the field is recognizing the full potential of the meningeal lymphatic vasculature as a novel and exciting therapeutic target for age-related diseases of the CNS,” Kipnis wrote to Alzforum (full comment below).
Still, researchers cautioned that it remains unclear how much Aβ is cleared through this versus other routes. “Several CSF clearance pathways have been described in rodents, including one along the olfactory nerve rootlets and cribriform plate, perineural pathways along the cranial nerves, and the dorsal and basal pathways discussed in this paper,” Costantino Iadecola at Weill Cornell Medical College, New York, wrote to Alzforum (full comment below). Roxana Carare and Roy Weller at the University of Southampton, U.K., wondered how meningeal lymphatics interact with interstitial fluid drainage along blood vessel walls, which they believe clears solutes and is associated with cerebral amyloid angiopathy (see comment below; Morris et al., 2016; Aldea et al., 2019).
Researchers also said the question of how immune cells commute between brain and periphery deserves more study. Nahrendorf reported that immune cells can travel directly from bone marrow inside the skull into brain tissue (Aug 2018 news). How do they get out? Kipnis noted that the dorsal meninges are loaded with immune cells. “Whilst the major route for CSF solutes may be through the basal lymphatics, the dural sinus is heavily populated by immune cells and their trafficking is happening throughout the entire meningeal lymphatic network, including dorsal sites,” he wrote (Louveau et al., 2018).—Madolyn Bowman Rogers
References
News Citations
- In Mice, CSF Caught Draining Via Lymphatic Vessels, Not Veins
- Lymphatic Vessels Found in Human Brain
- Lymphatic Brain Drain Withers in Aging, Worsens Disease
- Tiny Passageways Connect Skull Bone Marrow to the Brain
Research Models Citations
Paper Citations
- Ma Q, Ineichen BV, Detmar M, Proulx ST. Outflow of cerebrospinal fluid is predominantly through lymphatic vessels and is reduced in aged mice. Nat Commun. 2017 Nov 10;8(1):1434. PubMed.
- Choi I, Chung HK, Ramu S, Lee HN, Kim KE, Lee S, Yoo J, Choi D, Lee YS, Aguilar B, Hong YK. Visualization of lymphatic vessels by Prox1-promoter directed GFP reporter in a bacterial artificial chromosome-based transgenic mouse. Blood. 2011 Jan 6;117(1):362-5. Epub 2010 Oct 20 PubMed.
- Morris AW, Sharp MM, Albargothy NJ, Fernandes R, Hawkes CA, Verma A, Weller RO, Carare RO. Vascular basement membranes as pathways for the passage of fluid into and out of the brain. Acta Neuropathol. 2016 May;131(5):725-36. Epub 2016 Mar 14 PubMed.
- Aldea R, Weller RO, Wilcock DM, Carare RO, Richardson G. Cerebrovascular Smooth Muscle Cells as the Drivers of Intramural Periarterial Drainage of the Brain. Front Aging Neurosci. 2019;11:1. Epub 2019 Jan 23 PubMed.
- Louveau A, Herz J, Alme MN, Salvador AF, Dong MQ, Viar KE, Herod SG, Knopp J, Setliff JC, Lupi AL, Da Mesquita S, Frost EL, Gaultier A, Harris TH, Cao R, Hu S, Lukens JR, Smirnov I, Overall CC, Oliver G, Kipnis J. CNS lymphatic drainage and neuroinflammation are regulated by meningeal lymphatic vasculature. Nat Neurosci. 2018 Oct;21(10):1380-1391. Epub 2018 Sep 17 PubMed.
Further Reading
Primary Papers
- Ahn JH, Cho H, Kim JH, Kim SH, Ham JS, Park I, Suh SH, Hong SP, Song JH, Hong YK, Jeong Y, Park SH, Koh GY. Meningeal lymphatic vessels at the skull base drain cerebrospinal fluid. Nature. 2019 Jul 24; PubMed.
- Mäkinen T. Lymphatic vessels at the base of the mouse brain provide direct drainage to the periphery. Nature. 2019 Aug;572(7767):34-35. PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
Washington University in St. Louis, School of Medicine
Washington University in St Louis
Mayo Clinic
This is a beautiful paper, convincingly demonstrating that meningeal lymphatic vessels are indeed draining the CSF, whereas the perineural lymphatics are not. These results completely recapitulate our earlier findings (Louveau et al., 2015; Louveau et al., 2018; Da Mesquita et al., 2018) regarding the role of meningeal lymphatics in draining CSF solutes, brain ISF, and immune cells.
The authors elegantly demonstrate that lymphatic hot spots, sprout-like structures reminiscent of initial lymphatic vasculature, which we have previously described (Louveau et al., 2018), are indeed the access points for CSF drainage. The authors also present evidence that basal lymphatics are more active than dorsal lymphatics in CSF drainage and that this system dysfunctions with aging (as we showed in Da Mesquita et al., 2018).
Overall, the results reproduce our and others’ (Aspelund et al., 2015) findings on the role of meningeal lymphatics, put the role of perineural lymphatics in CSF drainage to rest, expand on the meningeal lymphatic network through basal lymphatics (which are a continuation of dorsal meningeal lymphatics), recapitulate the importance of lymphatic hot spots, and demonstrate their dysfunction in aging.
Whilst the major route for CSF solutes may be through the basal lymphatics, the dural sinus is heavily populated by immune cells and their trafficking is happening throughout the entire meningeal lymphatic network, including dorsal sites.
Targeting meningeal lymphatics alters Alzheimer’s disease progression (Da Mesquita et al., 2018). However, we do not know whether this is through a direct effect on CSF/β-amyloid drainage (happening through meningeal lymphatic network, presumably mainly through its basal part), via regulation of meningeal immunity (presumably mainly by dorsal lymphatics), or indeed both. These questions will need to be addressed to better understand how the meningeal lymphatic system, in its entirety, modulates CNS function and dysfunction. Step by step, the field is recognizing the full potential of the meningeal lymphatic vasculature as a novel and exciting therapeutic target for age-related diseases of the CNS.
References:
Louveau A, Smirnov I, Keyes TJ, Eccles JD, Rouhani SJ, Peske JD, Derecki NC, Castle D, Mandell JW, Lee KS, Harris TH, Kipnis J. Structural and functional features of central nervous system lymphatic vessels. Nature. 2015 Jul 16;523(7560):337-41. Epub 2015 Jun 1 PubMed.
Louveau A, Herz J, Alme MN, Salvador AF, Dong MQ, Viar KE, Herod SG, Knopp J, Setliff JC, Lupi AL, Da Mesquita S, Frost EL, Gaultier A, Harris TH, Cao R, Hu S, Lukens JR, Smirnov I, Overall CC, Oliver G, Kipnis J. CNS lymphatic drainage and neuroinflammation are regulated by meningeal lymphatic vasculature. Nat Neurosci. 2018 Oct;21(10):1380-1391. Epub 2018 Sep 17 PubMed.
Da Mesquita S, Louveau A, Vaccari A, Smirnov I, Cornelison RC, Kingsmore KM, Contarino C, Onengut-Gumuscu S, Farber E, Raper D, Viar KE, Powell RD, Baker W, Dabhi N, Bai R, Cao R, Hu S, Rich SS, Munson JM, Lopes MB, Overall CC, Acton ST, Kipnis J. Functional aspects of meningeal lymphatics in ageing and Alzheimer's disease. Nature. 2018 Aug;560(7717):185-191. Epub 2018 Jul 25 PubMed.
Aspelund A, Antila S, Proulx ST, Karlsen TV, Karaman S, Detmar M, Wiig H, Alitalo K. A dural lymphatic vascular system that drains brain interstitial fluid and macromolecules. J Exp Med. 2015 Jun 29;212(7):991-9. Epub 2015 Jun 15 PubMed.
Weill College Medicine, New York
The paper describes a lymphatic pathway for CSF clearance located at the base of the skull. It is remarkable that these vessels have the classical morphology and molecular signature of lymphatic vessels elsewhere in the body. The findings, in concert with the previously described dorsal lymphatic pathway along the sagittal sinus, further debunk the myth that the brain does not have functional lymphatic vessels.
Several CSF clearance pathways have been described in rodents, including one along the olfactory nerve rootlets and cribriform plate, perineural pathways along the cranial nerves, and the dorsal and basal pathways discussed in this paper. However, several issues remain to be addressed: Do these pathways exist in humans? What is their relative contribution to CSF clearance, i.e. is one pathway more important than another? Is the dorsal pathway, which has been confirmed in humans, draining only the meninges or also the CSF?
CSF production and clearance are reduced in aging, which has been implicated in the accumulation of toxic proteins in the brain (Aβ, tau, synuclein etc.). Which of these pathways is the most relevant to the disturbance in proteostasis caused by the CSF clearance deficit in aging remains to be established. Perhaps advanced imaging approaches could be used to probe this question in humans, and studies along these lines would provide much-needed clarity in this burgeoning field of research.
University of Southampton School of Medicine
University of Southampton School of Medicine
This is a very elegant anatomical and physiological study. It demonstrates the balance of the active meningeal lymphatics draining the CSF. It is particularly relevant, as rodent experimental models are the current model of choice for studies of drainage of interstitial and cerebrospinal fluids (Weller et al., 2018).
There are still aspects of the anatomy of the subarachnoid space and movement of CSF that are under investigation and are relevant to early diagnosis in Alzheimer’s disease (de Leon et al., 2017). It remains to be seen to what extent the basal meningeal lymphatics have a role in drainage of interstitial fluid of the brain along the intramural periarterial drainage (IPAD) pathways, especially as IPAD is impaired with vascular dysfunction that accompanies ageing (Sweeney et al., 2019; Hawkes et al., 2013).
References:
Weller RO, Sharp MM, Christodoulides M, Carare RO, Møllgård K. The meninges as barriers and facilitators for the movement of fluid, cells and pathogens related to the rodent and human CNS. Acta Neuropathol. 2018 Mar;135(3):363-385. Epub 2018 Jan 24 PubMed.
de Leon MJ, Li Y, Okamura N, Tsui WH, Saint-Louis LA, Glodzik L, Osorio RS, Fortea J, Butler T, Pirraglia E, Fossati S, Kim HJ, Carare RO, Nedergaard M, Benveniste H, Rusinek H. Cerebrospinal Fluid Clearance in Alzheimer Disease Measured with Dynamic PET. J Nucl Med. 2017 Sep;58(9):1471-1476. Epub 2017 Mar 16 PubMed.
Sweeney MD, Montagne A, Sagare AP, Nation DA, Schneider LS, Chui HC, Harrington MG, Pa J, Law M, Wang DJ, Jacobs RE, Doubal FN, Ramirez J, Black SE, Nedergaard M, Benveniste H, Dichgans M, Iadecola C, Love S, Bath PM, Markus HS, Salman RA, Allan SM, Quinn TJ, Kalaria RN, Werring DJ, Carare RO, Touyz RM, Williams SC, Moskowitz MA, Katusic ZS, Lutz SE, Lazarov O, Minshall RD, Rehman J, Davis TP, Wellington CL, González HM, Yuan C, Lockhart SN, Hughes TM, Chen CL, Sachdev P, O'Brien JT, Skoog I, Pantoni L, Gustafson DR, Biessels GJ, Wallin A, Smith EE, Mok V, Wong A, Passmore P, Barkof F, Muller M, Breteler MM, Román GC, Hamel E, Seshadri S, Gottesman RF, van Buchem MA, Arvanitakis Z, Schneider JA, Drewes LR, Hachinski V, Finch CE, Toga AW, Wardlaw JM, Zlokovic BV. Vascular dysfunction-The disregarded partner of Alzheimer's disease. Alzheimers Dement. 2019 Jan;15(1):158-167. PubMed. Correction.
Hawkes CA, Gatherer M, Sharp MM, Dorr A, Yuen HM, Kalaria R, Weller RO, Carare RO. Regional differences in the morphological and functional effects of aging on cerebral basement membranes and perivascular drainage of amyloid-β from the mouse brain. Aging Cell. 2013 Apr;12(2):224-36. PubMed.
University of Bern
Swiss Federal Institute of Technology (ETH) Zurich
This is an extremely interesting study that addresses a controversial and fundamental topic at the moment—how the cerebrospinal fluid (CSF) drains from the spaces around the brain and spinal cord. The textbook knowledge is that CSF drains directly into veins, however, this is primarily based on studies carried out over a century ago and has never been directly shown in an in vivo physiological study. In the last few years, work performed by a few groups including our own (Ma et al., 2017; Nov 2017 news) has highlighted the forgotten role that the lymphatics might be playing in this process. We showed that in mice, CSF clears predominantly through the lymphatic system rather than directly into blood. However, the anatomical route(s) that the fluid takes to reach lymphatic vessels are still controversial.
Ahn et al. propose that the meningeal lymphatic vessels located at the base of the skull (in the outermost dura mater layer of the linings of the central nervous system—not the brain itself) are the major route for CSF clearance. This is contradictory to the work performed by Jonathan Kipnis’ group at the University of Virginia, who have proposed that the meningeal lymphatic vessels on the dorsal side (top) of the skull are the most important. In agreement with the current study, our MRI data also support bulk CSF flow mostly in the basal, rather than dorsal, region (Ma et al., 2019).
In addition, our own group, backed up by dozens of historical studies (Koh et al., 2005), has shown that CSF outflow can also occur through routes along cranial nerves, especially in the nasal or optical regions, only reaching lymphatic vessels once these nerves have left the skull (Ma et al., 2017). These particular routes were not assessed in the current paper, so the question remains open which routes are the most important.
References:
Ma Q, Ineichen BV, Detmar M, Proulx ST. Outflow of cerebrospinal fluid is predominantly through lymphatic vessels and is reduced in aged mice. Nat Commun. 2017 Nov 10;8(1):1434. PubMed.
Ma Q, Ries M, Decker Y, Müller A, Riner C, Bücker A, Fassbender K, Detmar M, Proulx ST. Rapid lymphatic efflux limits cerebrospinal fluid flow to the brain. Acta Neuropathol. 2019 Jan;137(1):151-165. Epub 2018 Oct 10 PubMed.
Koh L, Zakharov A, Johnston M. Integration of the subarachnoid space and lymphatics: is it time to embrace a new concept of cerebrospinal fluid absorption?. Cerebrospinal Fluid Res. 2005 Sep 20;2:6. PubMed.
University of Louisville School of Medicine
This important study clarifies the role of meningeal lymphatic vessels in clearance of important molecules and immune cells from the brain to the periphery. This paper, and other work, have described how these molecules and cells go from the base of the brain to lymphatic meningeal vessels (Sweeney and Zlokovic, 2018). The lymphatic clearance pathway may be important in the pathogenesis of neurodegenerative diseases and other conditions. As Costantino Iadecola pointed out, other pathways may also be important, such as the olfactory nerve and the cribriform plate.
We must consider the full course of the passage of material from the brain to the reticuloendothelial system through the meningeal lymphatic vessels. As Ahn et al. propose, lymphatic clearance from the brain goes to the deep cervical lymph nodes. This process will likely be influenced by the oral, nasopharyngeal and laryngeal microbiota. We already know that the gut microbiota influences systemic and cerebral immune processes in profound ways (Lathrop et al., 2011; Rothhammer et al, 2018; Abdel-Haq et al., 2019). It is likely that microbiota in the head will similarly influence immune functions in the cervical lymph nodes to influence clearance processes. This may have important implications for stroke, vascular cognitive impairment, Alzheimer's disease, and idiopathic intracranial hypertension.
These relationships can be addressed in many ways in humans, including MRI and tonsillar biopsies, as well as studies of the relationship of CSF immune markers and oral bacteria.
References:
Abdel-Haq R, Schlachetzki JC, Glass CK, Mazmanian SK. Microbiome-microglia connections via the gut-brain axis. J Exp Med. 2019 Jan 7;216(1):41-59. Epub 2018 Nov 1 PubMed.
Rothhammer V, Borucki DM, Tjon EC, Takenaka MC, Chao CC, Ardura-Fabregat A, de Lima KA, Gutiérrez-Vázquez C, Hewson P, Staszewski O, Blain M, Healy L, Neziraj T, Borio M, Wheeler M, Dragin LL, Laplaud DA, Antel J, Alvarez JI, Prinz M, Quintana FJ. Microglial control of astrocytes in response to microbial metabolites. Nature. 2018 May;557(7707):724-728. Epub 2018 May 16 PubMed.
Sweeney MD, Zlokovic BV. A lymphatic waste-disposal system implicated in Alzheimer's disease. Nature. 2018 Aug;560(7717):172-174. PubMed.
Lathrop SK, Bloom SM, Rao SM, Nutsch K, Lio CW, Santacruz N, Peterson DA, Stappenbeck TS, Hsieh CS. Peripheral education of the immune system by colonic commensal microbiota. Nature. 2011 Sep 21;478(7368):250-4. PubMed.
Washington University in St. Louis, School of Medicine
Washington University in St Louis
Mayo Clinic
We would like to correct a comment made by Dr. Proulx and his colleague, which we consider misleading. The comment states:
“Ahn et al. propose that the meningeal lymphatic vessels located at the base of the skull (in the outermost dura mater layer of the linings of the central nervous system—not the brain itself) are the major route for CSF clearance. This is contradictory to the work performed by Jonathan Kipnis’ group at the University of Virginia, who have proposed that the meningeal lymphatic vessels on the dorsal side (top) of the skull are the most important. In agreement with the current study, our MRI data also support bulk CSF flow mostly in the basal, rather than dorsal, region (Ma et al., 2019).”
Since our rediscovery of the meningeal lymphatic vasculature in 2015, we have never divided it into “most important’ or “less important” zones. Supplemental Figure 1 of our recent paper (Louveau et al., Nat Neurosci 2018,) clearly demonstrates the role of basal lymphatics in CSF drainage. We have focused on the dorsal part of meningeal lymphatics because of its comparatively easier accessibility.
The work of Ahn et al., has recapitulated original findings from our lab (Louveau et al., Nature 2015; Louveau et al., Nat Neurosci 2018; Da Mesquita et al., 2018) and from the lab of Kari Alitalo (Aspelund et al, JEM 2015). It nicely showed that the basal part of the system, which is where dorsal vessels converge and drain into, is supposedly better equipped for CSF drainage than its dorsal part.
Interestingly, the previous work from Proulx’ lab (Ma et. al., Nat Comm 2017) claimed that the meningeal lymphatic vasculature has no role in CSF drainage, and that CSF is drained entirely through perineural routes. The current work by Ahn et al., clearly demonstrates that meningeal lymphatics indeed play a critical role in CSF drainage, as we and others have previously suggested.
The goal now is to focus our efforts on developing therapeutics to target meningeal lymphatic vessels in various neurological diseases.
Swiss Federal Institute of Technology (ETH) Zurich
University of Bern
We appreciate the new comment from Dr. Kipnis and colleagues and their willingness to engage in discussion on this controversial topic. We also feel the need to clarify some of the statements that have been made by these authors in their comments to this forum and to further put into context the exciting new findings by Ahn et al., 2019.
Dr. Kipnis and colleagues are correct that they may have never explicitly stated that the dorsal meningeal lymphatic vessels are “most important” for CSF clearance. However, it is difficult to interpret their recent data any other way. In the study by Louveau et al., 2018, after providing evidence supporting the functionality of the vessels, the authors utilized a photoablation technique that targeted the lymphatic vessels under the dorsal skull (as well as an additional small network of lymphatic vessels located in the dura near the cerebellum). As this method involved shining light through the intact skull to ablate the vessels, the procedure presumably left unaffected both the basal meningeal lymphatic vessels and the perineural outflow routes. The authors then proceeded to show dramatic reductions of CSF transport to draining lymph nodes of fluorescently labeled beads and T cells injected into the cisterna magna. They concluded from this data that the (ablated) meningeal lymphatic vessels are the “main route for immune cell and macromolecule drainage from the CSF.” The authors have indeed shown immunostainings of lymphatic vessels in the basal region of the skull in Supplemental Figure 2. However, they did not demonstrate functionality of these vessels and instead argued against this:
“After i.c.m. injection, some of the fluorescent particles are seen accumulating around the sides of the skull, close to lymphatic vessels (Supplementary Fig. 2j). These vessels, however, seemed to have lymphatic valves, as previously described (Aspelund et al., 2015), suggesting they were collecting lymphatics, which are usually less accessible by macromolecules.”
The current report by Ahn et al. has elegantly clarified the morphology of the lymphatic vessel network in the basal region, demonstrating the existence of blind-ended vessels and a patterning of the junctions between cells that would be more indicative of initial or precollecting lymphatic vessel network capable of draining fluid and solutes. Ahn et al. have also confirmed findings from our group that have indicated that the dorsal lymphatic network was discontinuous in nature and did not appear to participate in CSF tracer clearance (Ma et al., 2017). Thus, it is not immediately apparent how Dr. Kipnis and colleagues have come to the conclusion that the current report has “completely recapitulated” their earlier findings.
Regarding the claims made in our previous report, we invite Dr. Kipnis and colleagues to closely reread this paper (Ma et al., 2017). We have never claimed that the meningeal lymphatic vessels (we prefer the term dural lymphatic vessels, as a consensus has been reached that they are localized to the dura mater) (Mastorakos and McGavern, 2019) do not have a role in CSF drainage or that this fluid is drained entirely through perineural routes. In fact, in the discussion of this paper (Ma et al., 2017) we have written:
“Interestingly, anatomical studies have demonstrated lymphatic vessels in the dura around the optic and facial nerves and at the cribriform plate and jugular foramen, indicating that lymphatic uptake of CSF may occur as the cranial nerves exit the skull (Aspelund et al., 2015; Földi et al., 1966; Furukawa et al., 2008; Killer et al., 1999). Clearly, more research is needed to determine the importance of the dural lymphatic route for CSF outflow in comparison to the more established perineural pathways to reach extracranial lymphatic vessels.”
The current report by Ahn et al. has provided beautiful images that support data that we have published in collaboration with the group of Prof. Kari Alitalo at the University of Helsinki (Aspelund et al., 2015), in which tracers were detected within the dural lymphatic vessels at the base of the skull. We have also published data supporting an outflow of CSF-injected tracers from both the stylomastoid (where the facial nerve exits the skull) and jugular foramina (where the glossopharyngeal, vagus, and accessory nerves exit the skull), two of the routes identified in the current paper as outflow sites of dural lymphatic vessels from the cranium (Ma et al., 2017). The exact mechanism of fluid and solute uptake into the lymphatic network at these locations is still not clear. Ahn et al. have claimed that the arachnoid barrier layer is not intact in these regions, which could allow access of CSF to the interstitium of the dura and, thus, to the lymphatics of this tissue. This is a highly interesting finding that awaits further confirmation.
Finally, we feel the need to address the comments made by Dr. Kipnis and colleagues in their initial comment on July 26. The authors claim that the report by Ahn et al. has shown “meningeal lymphatic vessels are indeed draining the CSF, whereas the perineural lymphatics are not” and has “put the role of perineural lymphatics in CSF drainage to rest.” By “perineural lymphatics,” we presume that the authors are referring to CSF outflow routes along cranial nerves as they exit the skull. If this presumption is correct, then these statements have no basis in fact. The new report did not assess these routes, as we have pointed out in our initial comment. The evidence in the literature for CSF egress in several species through foramina at the cribriform plate (where olfactory nerves exit the skull) and through the optic foramina (where the optic nerve exits) is very strong. While the exact access points along these routes to lymphatic vessels remains a point of contention, tracers injected into the CSF can be routinely demonstrated by imaging techniques to be within collecting lymphatic vessels outside the skull of these regions (e.g., Ma et al., 2017).
This area of research is currently very active, with several independent groups pursuing studies that aim to elucidate the mechanisms of CSF flow and clearance from the central nervous system. Let’s all avoid making premature conclusions before all the evidence can be weighed. We can take strides to engage in an open-minded and constructive discourse between labs. This will facilitate progress toward our shared goal of translating these exciting new discoveries into therapeutic targets for the treatment of neurodegenerative disorders and other CNS diseases.
References:
Aspelund A, Antila S, Proulx ST, Karlsen TV, Karaman S, Detmar M, Wiig H, Alitalo K. A dural lymphatic vascular system that drains brain interstitial fluid and macromolecules. J Exp Med. 2015 Jun 29;212(7):991-9. Epub 2015 Jun 15 PubMed.
Földi M, Gellért A, Kozma M, Poberai M, Zoltán OT, Csanda E. New contributions to the anatomical connections of the brain and the lymphatic system. Acta Anat (Basel). 1966;64(4):498-505. PubMed.
Furukawa M, Shimoda H, Kajiwara T, Kato S, Yanagisawa S. Topographic study on nerve-associated lymphatic vessels in the murine craniofacial region by immunohistochemistry and electron microscopy. Biomed Res. 2008 Dec;29(6):289-96. PubMed.
Killer HE. Production and circulation of cerebrospinal fluid with respect to the subarachnoid space of the optic nerve. J Glaucoma. 2013 Jun-Jul;22 Suppl 5:S8-10. PubMed.
Louveau A, Herz J, Alme MN, Salvador AF, Dong MQ, Viar KE, Herod SG, Knopp J, Setliff JC, Lupi AL, Da Mesquita S, Frost EL, Gaultier A, Harris TH, Cao R, Hu S, Lukens JR, Smirnov I, Overall CC, Oliver G, Kipnis J. CNS lymphatic drainage and neuroinflammation are regulated by meningeal lymphatic vasculature. Nat Neurosci. 2018 Oct;21(10):1380-1391. Epub 2018 Sep 17 PubMed.
Ma Q, Ineichen BV, Detmar M, Proulx ST. Outflow of cerebrospinal fluid is predominantly through lymphatic vessels and is reduced in aged mice. Nat Commun. 2017 Nov 10;8(1):1434. PubMed.
Mastorakos P, McGavern D. The anatomy and immunology of vasculature in the central nervous system. Sci Immunol. 2019 Jul 12;4(37) PubMed.
Discussion of the discovery of lymph vessels in the dura should not ignore the work of Andres et al. (1987). They showed an e.m. with two lymph vessels, and a drawing illustrating their relation to blood vessels and nerves. They included a full paragraph describing them, which begins: “The dura mater encephali carries lymphatic capillaries. They occur around the wall of the sagittal sinus, in the area of the confluence of sinuses in close contact to the mesothelial cells of the subdural space, and in the vicinity of the vascular bed of the dural tissue”
They also write: “Their functional significance is not known. They may serve to drain the subarchnoidal space.” Admittedly, this is in dogs and cats, not mice. Somewhat earlier, Waggener and Beggs (1967) stated that in the dura of rats and hamsters, “blood vessels, lymphatics, and nerve fasculi are grouped in focal aggregates,” but did not show this in a micrograph.
References:
Andres KH, von Düring M, Muszynski K, Schmidt RF. Nerve fibres and their terminals of the dura mater encephali of the rat. Anat Embryol (Berl). 1987;175(3):289-301. PubMed.
Waggener JD, Beggs J. The membranous coverings of neural tissues: an electron microscopy study. J Neuropathol Exp Neurol. 1967 Jul;26(3):412-26. PubMed.
Make a Comment
To make a comment you must login or register.