Lymphatic Vessels Found in Human Brain
Quick Links
For decades, the lymphatic system that removes waste and transports white blood cells around the body was considered absent from the mammalian brain. Then in 2015, groups in Finland and the United States independently reported the existence of an active lymphatic system in the outermost layer of brain covering, the dura mater. That was in mice. Now, a paper in the October 3 eLife extends the work to people and marmoset monkeys. Researchers led by Daniel Reich at the National Institute of Neurological Disorders and Stroke, Bethesda, Maryland, tweaked MRI protocols to allow the scans to light up lymphatic vessels in the dura. Immunostaining on postmortem brains confirmed that these structures were indeed lymphatic and not blood vessels. The system may represent a previously unknown clearance route for immune cells and large molecules from the human brain, although that remains to be demonstrated, Reich said. If so, the discovery would alter fundamental assumptions. “The communication between the immune system and the human brain may not be as different from the rest of the body as we initially thought,” he told Alzforum.
- Recent mouse studies upended the belief that the brain lacks a lymphatic system.
- Now researchers have visualized lymphatic vessels in living human brains.
- It is unknown if the system plays a role in disease.
Other researchers welcomed the new ability to image these vessels in living people. “The work reported in this paper is a major step forward in the study of dural lymphatics … In vivo imaging in patients would greatly enhance our knowledge of the pathophysiology of intractable diseases such as multiple sclerosis and dementia, and may aid in their management and therapy,” Roy Weller and Roxana Carare at the University of Southampton, England, U.K., wrote to Alzforum (see full comment below).
The presence of these lymphatic vessels in the membranes, i.e., meninges, that wrap the central nervous system had been noted before, with the Italian anatomist Paolo Mascagni, credited with discovery of the lymph system, first reporting their existence in 1787 (Bucchieri et al., 2015; Mascagni 1787). More recent studies also described structures in the human dura that could be lymphatic vessels (Li et al., 1996). Others dismissed these as blood vessels, which are easily confused with lymph vessels.
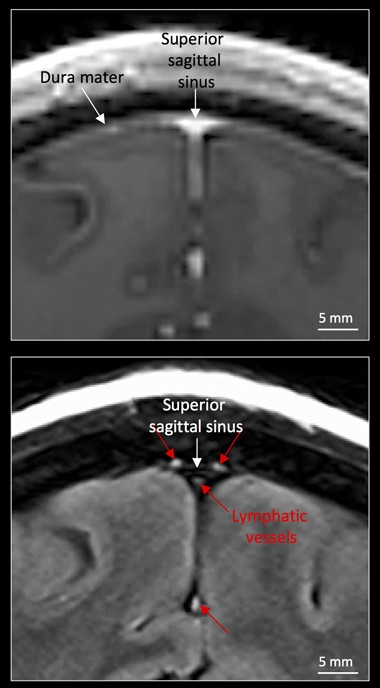
Brain Lymph.
On normal MRI scans (top), bright blood vessels in the dura obscure lymphatic vessels, but when the signal from blood is tuned out (bottom), the latter become visible (red arrows). [Courtesy of Absinta et al., eLife.]
Specific markers for lymphatic vessels were identified only recently, and the tough, leathery dura mater resists staining, Reich noted. Still, interest in the topic of brain lymph surged after the mouse studies revealed a functional lymphatic system in mammalian meninges. In mice, the system cleared cerebrospinal fluid, large molecules, and immune cells from the brain and transported them to cervical lymph nodes (Aspelund et al., 2015; Louveau et al., 2015).
Those findings inspired Reich to look for lymphatic vessels in human brains using MRI. To visualize meningeal vessels, joint first authors Martina Absinta and Seung-Kwon Ha used the gadolinium-based dye gadobutrol. Because blood vessels in the dura lack a blood-brain barrier, contrast agents such as gadobutrol can leak out and be taken up by adjacent lymphatic vessels. However, the signal from any such vessels is normally swamped by the signal from blood, which produces a bright smear throughout the dura on MRI scans. To get around this, the researchers tuned the scanner to subtract the blood signal, in effect rendering these vessels invisible. This is possible because blood flows faster and affects the magnetic properties of gadolinium differently than lymph, Reich noted. With the bright blood vessels blotted out, small surrounding vessels became visible (see image above). These measured about 1 millimeter in diameter, and were present in all five people examined. The vessels ran parallel to the dural venous sinuses, which drain blood from the brain, and matched the lymphatic arrangement reported in mice.
The researchers also scanned three marmosets, and saw similar vessels light up with gadobutrol. On pathological examination of the marmoset brains, the vessels expressed lymphatic markers. The authors immunostained three human postmortem brains for lymphatic markers, as well. The technique revealed a branched network of lymphatic vessels in the dura, including many small vessels only a few microns in diameter that were invisible on MRI. Reich noted that it took some time for his group to find the right staining conditions for the dura, perhaps explaining why these lymphatic vessels had not been identified before.
Reich’s study did not examine how the lymphatic vessels worked. Costantino Iadecola at Weill Cornell Medical College in New York noted that it remains to be seen if they drain fluid and solutes from the brain parenchyma, as the mouse vessels do. It is also unclear how this lymphatic system interacts with the “glymphatic” drainage system identified in the human brain, in which astrocytes help drive fluid flow through brain parenchyma, carrying away soluble Aβ and other waste products (Aug 2012 news; Mar 2013 news; Dec 2016 news). Reich believes that the glymphatic fluid that helps rinse the brain parenchyma may eventually end up in the lymphatic channels of the dura, although more research will be needed to find out how these systems connect.
Look—Lymphatics! MRI reveals lymphatic vessels (green) in the dura of a healthy 47-year-old woman. [Courtesy of Absinta et al., eLife.]
Iadecola was particularly intrigued by the implications of this lymphatic system for brain immune function. Researchers used to think the brain had but a limited immune response. That’s because white blood cells can enter the brain and be exposed to antigens there, but there was no known pathway for them to leave. If these cells slip out through lymphatic channels, however, then they could travel to lymphoid organs, proliferate, and return to the brain in force to trigger a dramatic immune response. Iadecola wondered if this could be a factor in multiple sclerosis, where immune cells attack the white matter of the brain. He noted that scientists now know the meninges are loaded with lymphocytes, which can enter the brain after injury (Kim et al., 2009; Roth et al., 2014). “The meninges are emerging as almost a lymphoid organ, so it would make sense that there is this lymphatic pathway in the meninges to maintain homeostasis,” Iadecola said.
Others speculated on the applications for neurodegenerative disease. “The meningeal lymphatic system could also play a role in removing metabolic waste and proteinaceous toxic accumulates from brain. It would be very interesting to determine whether there is any dysfunction in lymphatic drainage in neurological diseases, especially neurodegenerative diseases which have aberrant protein accumulation,” Berislav Zlokovic and colleagues at the University of Southern California in Los Angeles wrote to Alzforum (see full comment below).
Reich plans to study whether meningeal lymphatic vessels malfunction in various neurological and neurodegenerative diseases. He noted that the MRI methods used in this study are variations on ones used routinely in clinical practice, and easily could be applied to disease studies.—Madolyn Bowman Rogers
References
News Citations
- Brain Drain—“Glymphatic” Pathway Clears Aβ, Requires Water Channel
- Spinal Fluid Flush: Visualizing the Brain Drain With MRI
- Dearth of Water Channels a Sign of ‘Glymphatic’ Breakdown in Alzheimer’s?
Paper Citations
- Bucchieri F, Farina F, Zummo G, Cappello F. Lymphatic vessels of the dura mater: a new discovery?. J Anat. 2015 Nov;227(5):702-3. Epub 2015 Sep 18 PubMed.
- Li J, Zhou J, Shi Y. Scanning electron microscopy of human cerebral meningeal stomata. Ann Anat. 1996 Jun;178(3):259-61. PubMed.
- Aspelund A, Antila S, Proulx ST, Karlsen TV, Karaman S, Detmar M, Wiig H, Alitalo K. A dural lymphatic vascular system that drains brain interstitial fluid and macromolecules. J Exp Med. 2015 Jun 29;212(7):991-9. Epub 2015 Jun 15 PubMed.
- Louveau A, Smirnov I, Keyes TJ, Eccles JD, Rouhani SJ, Peske JD, Derecki NC, Castle D, Mandell JW, Lee KS, Harris TH, Kipnis J. Structural and functional features of central nervous system lymphatic vessels. Nature. 2015 Jul 16;523(7560):337-41. Epub 2015 Jun 1 PubMed.
- Kim JV, Kang SS, Dustin ML, McGavern DB. Myelomonocytic cell recruitment causes fatal CNS vascular injury during acute viral meningitis. Nature. 2009 Jan 8;457(7226):191-5. Epub 2008 Nov 16 PubMed.
- Roth TL, Nayak D, Atanasijevic T, Koretsky AP, Latour LL, McGavern DB. Transcranial amelioration of inflammation and cell death after brain injury. Nature. 2014 Jan 9;505(7482):223-8. Epub 2013 Dec 8 PubMed.
External Citations
Further Reading
Primary Papers
- Absinta M, Ha SK, Nair G, Sati P, Luciano NJ, Palisoc M, Louveau A, Zaghloul KA, Pittaluga S, Kipnis J, Reich DS. Human and nonhuman primate meninges harbor lymphatic vessels that can be visualized noninvasively by MRI. Elife. 2017 Oct 3;6 PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
University of Southampton School of Medicine
University of Southampton School of Medicine
This manuscript strengthens the data observed in rodents and described in Louveau et al. (2015). Using T2-FLAIR and T1-weighted black-blood imaging in humans and marmoset monkeys, the dural lymphatic vessels were observed in the present work bilaterally along the superior sagittal sinus, and immunocytochemistry for lymphatic markers demonstrated their presence in dura mater.
The German neuroanatomist Karl-Hermann Andres (1929-2005, Ruhr-Universität Bochum) published in 1987 that:
Lymphatic vessels occur within the dura mater. They leave the cranial cavity through the openings of the cribriform plate, rostral to the bulla tympani together with the transverse sinus, and the middle meningeal artery.
There is firm evidence that lymphatic drainage of the cerebrospinal fluid is along different anatomical compartments compared to that of the cerebral parenchyma, resulting in distinct implications in neuroimmunology (Engelhardt et al., 2016). Some parenchymal interstitial fluid escapes into the CSF and will most likely reach the dural lymphatic vessels (McIntee et al., 2016; Szentistvanyi et al., 1984).
The work reported in this paper is a major step forward in the study of dural lymphatics. Although much can be discovered by histology and tracer studies in experimental animals, visualizing dural lymphatics by imaging in humans allows dural lymphatics to be studied clinically in neurological diseases ranging from multiple sclerosis to dementia. Many obstacles remain before there is a full understanding of the functions of dural lymphatics in relation to the central nervous system. The dura is separated from the CSF in the subarachnoid space by a waterproof, impenetrable layer of arachnoid. The first question therefore is how CSF reaches dural lymphatics, which it undoubtedly does. Once this problem has been solved, it may be possible to expand the use of imaging of dural lymphatics to study the dynamics of CSF drainage by this route and how it is altered in neurological disease.
The second question that remains to be resolved is the relationship between dural lymphatics and lymphatic drainage of the brain. Studies involving the injection of tracers into the brain itself show that lymphatic drainage pathways are along the walls of cerebral capillaries and arteries. This is supported by observations in the human brain whereby amyloid is deposited in the intramural peri-arterial drainage (IPAD) pathways as cerebral amyloid angiopathy (Morris et al., 2016).
These are exciting times, with the promise of visualising routes of lymphatic drainage by MRI not only in the dura but in the central nervous system. In vivo imaging in patients would greatly enhance our knowledge of the pathophysiology of intractable diseases such as multiple sclerosis and dementia and may aid in their management and therapy.
References:
Louveau A, Smirnov I, Keyes TJ, Eccles JD, Rouhani SJ, Peske JD, Derecki NC, Castle D, Mandell JW, Lee KS, Harris TH, Kipnis J. Structural and functional features of central nervous system lymphatic vessels. Nature. 2015 Jul 16;523(7560):337-41. Epub 2015 Jun 1 PubMed.
Engelhardt B, Carare RO, Bechmann I, Flügel A, Laman JD, Weller RO. Vascular, glial, and lymphatic immune gateways of the central nervous system. Acta Neuropathol. 2016 Sep;132(3):317-38. Epub 2016 Aug 13 PubMed.
McIntee FL, Giannoni P, Blais S, Sommer G, Neubert TA, Rostagno A, Ghiso J. In vivo Differential Brain Clearance and Catabolism of Monomeric and Oligomeric Alzheimer's Aβ protein. Front Aging Neurosci. 2016;8:223. Epub 2016 Sep 27 PubMed.
Szentistványi I, Patlak CS, Ellis RA, Cserr HF. Drainage of interstitial fluid from different regions of rat brain. Am J Physiol. 1984 Jun;246(6 Pt 2):F835-44. PubMed.
Morris AW, Sharp MM, Albargothy NJ, Fernandes R, Hawkes CA, Verma A, Weller RO, Carare RO. Vascular basement membranes as pathways for the passage of fluid into and out of the brain. Acta Neuropathol. 2016 May;131(5):725-36. Epub 2016 Mar 14 PubMed.
University of South Alabama
University of Edinburgh
University of Southern California, Keck School of Medicine
University of Southern California
In 1787, anatomist Paolo Mascagni published an atlas of all lymphatic vessels in the human body, which included the brain, where he described the presence of lymphatics in the cerebral dura mater (Mascagni, 1787, Vasorum Lymphaticorum Corporis Humani Historia et Ichonographia). In the late 19th century, Gustav Schwalbe injected Prussian blue into the subarachnoid space and found that cervical lymph vessels were stained (Schwalbe, 1869, Der Arachnoidealraum, ein Lymphraum und sein Zusammenhang mit dem Perichorioidealraum). Throughout the 20th century, there were many studies investigating brain lymphatic system drainage. The Fӧldi group described the presence of “prelymphatics” in the brain, as they obstructed the lymphatic vessels in the necks of several animals, including dog, cat, and rabbit, and observed edema in the brains and enlargement of perivascular spaces (Casley-Smith et al., 1976; Csanda et al., 1963, 1968; Földi et al., 1966). Furthermore, studies using intracerebral injections of radioiodinated albumin into rabbit caudate nucleus (Bradbury et al., 1981) and large molecular weight tracers (India ink or albumin labeled with colloidal gold, Evans blue, or rhodamine) into the perivascular space of an artery on the brain surface, or within the cerebral cortices or the subarachnoid spaces of anesthetized rats (Ichimura et al., 1991), demonstrated that solutes carried by the perivascular interstitial fluid (ISF) flow reach the cerebrospinal fluid (CSF) compartment and drain into deep cervical lymph (Bradbury et al., 1981; Ichimura et al., 1991).
Two recent studies revealed the presence and role of the meningeal lymphatic vascular system in clearance of ISF and macromolecules from the mouse brain into the deep cervical lymph nodes (Aspelund et al., 2015; Louveau et al., 2015). Using Prox1-GFP transgenic mice, and injection of an inert 20-kDa poly(ethylene glycol) conjugate of the bright near-infrared dye IRDye 680 into brain parenchyma, Aspelund et al. tracked dural lymphatic vessels and the drainage of the dye into the deep cervical lymph nodes. Furthermore, they found that a transgenic mouse model with impaired vascular endothelial growth factor-C/D–vascular endothelial growth factor receptor 3 signaling lack dural lymphatic vessels, and have impaired clearance of macromolecules from brain into deep cervical lymph nodes (Aspelund et al., 2015). Louveau et al. identified several molecular hallmarks of mouse lymphatic endothelial cells in a whole-mount dissection of the dura mater, and also performed in vivo functional studies tracking flow of dye through lymphatic vessels by simultaneously injecting adult mice with fluorescein intravenously and with fluorescent tracer dye (QDot655) intracerebroventricularly, and imaging by multiphoton microscopy through a thinned skull. Furthermore, they identified vessels expressing similar lymphatic endothelial markers (Lyve-1+podoplanin+CD68–) by immunohistochemistry in postmortem human brain dura.
These previous studies demonstrated that the brain indeed has a lymphatic system, which here Absinta et al. confirmed in living brains in five young healthy human volunteers and three healthy adult non-human primates (marmosets) using magnetic resonance imaging (MRI) with a combination of regular Gadolinium-based contrast agent (Gadovist) and blood-pool contrast agent (Vasovist). Indeed, on T2-weighted fluid-attenuated inversion recovery (FLAIR) and T1-weighted black-blood imaging, lymphatic vessels enhance with Gadovist, but not with Vasovist. They also confirmed the vessels imaged by MRI to be lymphatic by immunohistochemistry using classical lymphatic endothelial markers (i.e., lymphatic vessel endothelial hyaluronan receptor 1 [LYVE-1], podoplanin [D2-40], prospero homeobox protein 1 [PROX1], COUP transcription factor 2 [COUP-TFII], and CCL21) versus vascular endothelial marker CD31 in both humans and marmosets.
In addition to its potential role in regulating brain immune responses, the meningeal lymphatic system could also play a role in removing metabolic waste and proteinaceous toxic accumulates from brain. It would be very interesting to determine whether there is any dysfunction in lymphatic drainage in neurological diseases, especially neurodegenerative diseases which have aberrant protein accumulation. For example, recent work from Kovacs et al. reported amyloid accumulation in the dura of Creutzfeldt-Jakob disease patients using postmortem histology (Kovacs et al., 2016), which in light of the present findings hints at the possibility of amyloid accumulation by or due to dysfunction of lymphatic vessels. Although the majority of Aβ is cleared from the murine brain under physiological conditions by transvascular receptor-mediated transport across the blood-brain barrier via clearance receptors including low-density lipoprotein receptor-related protein 1 (LRP1) and P-glycoprotein (PgP), about 15–20 percent is cleared from brain by perivascular flow of ISF (Shibata et al., 2000; Xie et al., 2013), likely draining into meningeal lymphatic vessels and deep cervical lymph nodes. Since in Alzheimer’s disease transvascular clearance systems fail relatively early due to damage of blood vessels associated with low expression of LRP1 and PgP (Montagne et al., 2017), it would be interesting to determine if the perivascular Aβ clearance system is also disrupted, leading to accumulation of Aβ along arterial blood vessels and within the lymphatic system. Moreover, whether the perivascular lymphatic system can be used to drain Aβ from brain remains an open question.
References:
Aspelund A, Antila S, Proulx ST, Karlsen TV, Karaman S, Detmar M, Wiig H, Alitalo K. A dural lymphatic vascular system that drains brain interstitial fluid and macromolecules. J Exp Med. 2015 Jun 29;212(7):991-9. Epub 2015 Jun 15 PubMed.
Bradbury MW, Cserr HF, Westrop RJ. Drainage of cerebral interstitial fluid into deep cervical lymph of the rabbit. Am J Physiol. 1981 Apr;240(4):F329-36. PubMed.
Casley-Smith JR, Földi-Börsök E, Földi M. The prelymphatic pathways of the brain as revealed by cervical lymphatic obstruction and the passage of particles. Br J Exp Pathol. 1976 Apr;57(2):179-88. PubMed.
CSANDA E, ZOLTAN TO, FOLDI M. Elevation of cerebrospinal-fluid pressure in the dog after obstruction of cervical lymphatic channels. Lancet. 1963 Apr 13;1(7285):832. PubMed.
Csanda E, Földi M, Obál F, Zoltán OT. Cerebral oedema as a consequence of experimental cervical lymphatic blockage. Angiologica. 1968;5(1):55-63. PubMed.
Földi M, Gellért A, Kozma M, Poberai M, Zoltán OT, Csanda E. New contributions to the anatomical connections of the brain and the lymphatic system. Acta Anat (Basel). 1966;64(4):498-505. PubMed.
Ichimura T, Fraser PA, Cserr HF. Distribution of extracellular tracers in perivascular spaces of the rat brain. Brain Res. 1991 Apr 5;545(1-2):103-13. PubMed.
Kovacs GG, Lutz MI, Ricken G, Ströbel T, Höftberger R, Preusser M, Regelsberger G, Hönigschnabl S, Reiner A, Fischer P, Budka H, Hainfellner JA. Dura mater is a potential source of Aβ seeds. Acta Neuropathol. 2016 Jun;131(6):911-23. Epub 2016 Mar 25 PubMed.
Louveau A, Smirnov I, Keyes TJ, Eccles JD, Rouhani SJ, Peske JD, Derecki NC, Castle D, Mandell JW, Lee KS, Harris TH, Kipnis J. Structural and functional features of central nervous system lymphatic vessels. Nature. 2015 Jul 16;523(7560):337-41. Epub 2015 Jun 1 PubMed.
Shibata M, Yamada S, Kumar SR, Calero M, Bading J, Frangione B, Holtzman DM, Miller CA, Strickland DK, Ghiso J, Zlokovic BV. Clearance of Alzheimer's amyloid-ss(1-40) peptide from brain by LDL receptor-related protein-1 at the blood-brain barrier. J Clin Invest. 2000 Dec;106(12):1489-99. PubMed.
Xie L, Kang H, Xu Q, Chen MJ, Liao Y, Thiyagarajan M, O'Donnell J, Christensen DJ, Nicholson C, Iliff JJ, Takano T, Deane R, Nedergaard M. Sleep drives metabolite clearance from the adult brain. Science. 2013 Oct 18;342(6156):373-7. PubMed.
Montagne A, Zhao Z, Zlokovic BV. Alzheimer's disease: A matter of blood-brain barrier dysfunction?. J Exp Med. 2017 Nov 6;214(11):3151-3169. Epub 2017 Oct 23 PubMed.
Make a Comment
To make a comment you must login or register.