Tau Propagation Surprise: It Might Travel Retrogradely
Quick Links
According to a leading theory, pathologic tau creeps through the brain by following synaptic connections. Evidence for this idea is still indirect. In a preprint posted to bioRXiv May 24, researchers led by Marc Diamond at University of Texas Southwestern Medical Center, Dallas, in collaboration with Julian Meeks at the University of Rochester Medical School, New York, and Julie Harris at the Allen Institute for Brain Science in Seattle, Washington, strengthened the case for network propagation of tau. Surprisingly, they report that the protein moves in the opposite direction from what was expected.
- An anti p-tau nanobody offers a 3-D look at propagation in mouse brain.
- In tauopathy mice, deposits arose spontaneously in several subcortical regions.
- Propagation followed synaptic connections—but retrogradely, not anterogradely.
The scientists labeled p-tau in whole brains from tauopathy mice using a single variable domain antibody, also called a nanobody because of its small size. These are only produced by a few animals, such as llamas. This small antibody was able to penetrate throughout the mouse brain, providing a high-resolution, three-dimensional view of where p-tau clustered. Comparing p-tau deposits in mice of different ages, the researchers calculated that network propagation, rather than diffusion, best explained the distribution patterns. The models turned up a surprise, however—the patterns fit better with retrograde, rather than anterograde, axonal transport. If the finding holds up, researchers might have to rethink their models for how pathologic tau spreads between neurons, Diamond said.
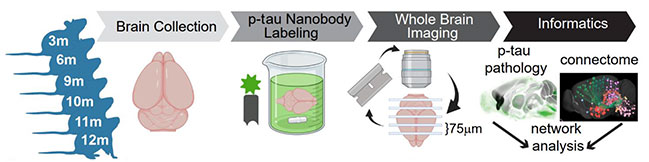
Capturing Whole-Brain Pathology. A new method labels whole brains from different age mice (left) by incubating them with an anti-p-tau nanobody (green), then examining thick blocks via serial two-photon tomography, and finally comparing findings against a connectivity atlas (right). [Courtesy of Ramirez et al., 2023.]
Human tau PET studies imply that tangles spread through connected brain regions, but these studies are necessarily low-resolution (Jan 2020 news; May 2020 news; Nov 2020 news). Mouse studies take a more granular look, but usually require injecting pathologic tau, rather than examining endogenous pathology. In addition, these experiments use thin brain slices that cannot provide a three-dimensional view.
To pinpoint endogenous tau more precisely and broadly, Diamond and colleagues turned to a nanobody previously generated at France’s Institut Pasteur. VHH-A2-488 recognizes p-tau422 and labels the same deposits as the well-known AT8 that binds p-tau202/205 (Li et al., 2016). Co-first author Denise Ramirez at UT Southwestern took intact, fixed brains from PS19 mice, which express mutant human P301S tau, permeabilized them using a detergent soak, then incubated them with VHH-A2-488 for six days. The labeled brains were cut into thick blocks and imaged with serial two-photon tomography to locate p-tau422. Finally, co-first authors Jennifer Whitesell and Nikhil Bhagwat at the Allen Brain Institute lined up these 3-D images with their connectivity atlas to achieve cellular resolution of tau pathology.
With this method, the authors compared brains from 28 mice of six different ages—three, six, nine, 10, 11, and 12 months. At six months, sparse tau deposits clustered in several distinct subcortical regions, as though they arose spontaneously in each. The seven with the most pathology were the locus ceruleus, anterior hypothalamic nucleus, posterior amygdalar nucleus, medial vestibular nucleus, Barrington’s nucleus, entorhinal area medial part dorsal zone, and the paragigantocellular reticular nucleus lateral part. At later ages, tau deposits became more widespread and contiguous.
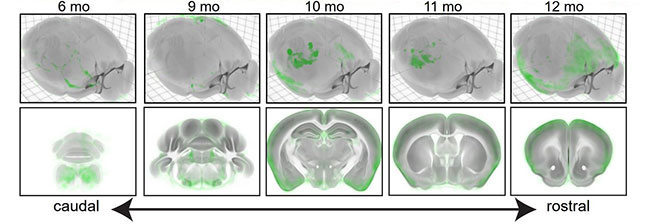
Encroaching Pathology. P-tau deposits (green) in tauopathy mice steadily increase with age, starting at the back of the brain at young ages and traveling forward (bottom). [Courtesy of Ramirez et al., 2023.]
Evaluating different models of spread, the authors found the data poorly fit a model based on proximity alone, suggesting p-tau does not simply diffuse through the brain. Instead, deposits seemed to follow brain connections. Surprisingly, the deposition pattern correlated only weakly with the strength of anterograde signaling between regions. The model produced a much better fit with the observed patterns of distribution when it assumed tau trafficked retrogradely up axons. A bi-directional model gave the second-best fit.
Retrograde trafficking is not unusual in pathology. Many toxins, such as rabies virus, are trafficked retrogradely. William Seeley at the University of California, San Francisco, speculated that because tau mis-localizes into dendrites in Alzheimer’s, and other diseases, that it may jump from post- to pre-synapse, where it is then trafficked down axons.
A second surprise was that three of the mice, all 10 months of age or older, harbored a different pattern of tau deposition than the others. Their deposits were predominantly cortical (image below). Diamond noted this pattern could represent a variant form of disease. In Alzheimer’s and other tauopathies, some people have distinct patterns of brain atrophy and develop different clinical symptoms than the norm (Apr 2021 news).

Two Patterns of Pathology. In the brains of tauopathy mice that are 10 months or older, p-tau (purple) typically deposits in subcortical regions (bottom). However, a few mice have predominantly cortical p-tau (top). [Courtesy of Ramirez et al., 2023.]
In future work, Diamond will use nanobodies specific for aggregated tau to look for any differences in the propagation pattern compared with anti-p-tau422. He noted the nanobody technique could be used to track other proteins of interest, or to investigate more complicated questions such as the relationship between tau deposition and microglial activation.—Madolyn Bowman Rogers.
References
News Citations
- Connectivity, Not Proximity, Predicts Tau Spread
- Simulating Tangle Spread Along Axons, Scientists Predict Tau PET Patterns
- Individualized Tau PET Model Outperforms Predictive Power of Braak Staging
- Forget Typical Alzheimer's: AI Finds Four Types.
Research Models Citations
Paper Citations
Further Reading
News
- What Drives Tangles to Spread? Answers Start Rolling In
- Abnormal Tau Slips into Synapses Long Before Tangles Form
- Bassoon Heralds Spread of Toxic Tau
- Doubling of Tau Seeds, Not Spread, Sets Pace of Tauopathy in Alzheimer's
- Does Fast-Progressing Alzheimer's Have a Whole Repertoire of Tau Conformers?
- Viral Proteins Help Shuttle Tau Aggregates Among Cells
- Wolframin-1 Cells: Tau’s Launch Pad from Entorhinal Cortex to Hippocampus?
- Do Lysosomes Help Propagate Tau Seeds?
Primary Papers
- Ramirez DM, Whitesell JD, Bhagwat N, Thomas TL, Ajay AD, Nawaby A, Delatour B, Bay S, Lafaye P, Knox JE, Harris JA, Meeks JP, Diamond MI. Endogenous pathology in tauopathy mice progresses via brain networks. 2023 May 24 10.1101/2023.05.23.541792 (version 1) bioRxiv.
Annotate
To make an annotation you must Login or Register.

Comments
Cliniques Universitaires Saint-Luc and Massachusetts General Hospital
Université Catholique de Louvain
This study explores the mechanisms of tau propagation through brain networks, using whole-brain staining methods with anti-p-tau nanobodies in 3D-imaged, P301S tau-mutated mice from 3 to 12 months old. The authors applied network propagation modeling to assess the relationship between structural connectivity and patterns of tau deposition across the whole brain. With their model, Denise Ramirez and colleagues report characteristic and non-random patterns of tau deposition over time, with a bias toward retrograde propagation.
These results are concordant with previous functional and diffusion-weighted MRI studies suggesting that tau pathology follows connected regions in humans (Adams et al., 2019; Franzmeier et al., 2020; Jacobs et al., 2018). However, the limited spatial resolution of PET data constrains the mapping precision of human tau deposition in vivo. In addition, neuroimaging studies in humans are unable to determine the direction of propagation, i.e., anterograde versus retrograde.
At a spatial resolution currently unthinkable at the human level, the results obtained through this innovative pipeline confirm that tau pathology preferentially propagates to structurally connected regions. This raises the question of the complementarity between diffusion-weighted and functional MRI techniques as predictors of tau propagation in humans.
Future longitudinal studies comparing the two modalities would be useful to better approach their respective associations with tau spreading.
References:
Adams JN, Maass A, Harrison TM, Baker SL, Jagust WJ. Cortical tau deposition follows patterns of entorhinal functional connectivity in aging. Elife. 2019 Sep 2;8 PubMed.
Franzmeier N, Neitzel J, Rubinski A, Smith R, Strandberg O, Ossenkoppele R, Hansson O, Ewers M, Alzheimer’s Disease Neuroimaging Initiative (ADNI). Functional brain architecture is associated with the rate of tau accumulation in Alzheimer's disease. Nat Commun. 2020 Jan 17;11(1):347. PubMed.
Jacobs HI, Hedden T, Schultz AP, Sepulcre J, Perea RD, Amariglio RE, Papp KV, Rentz DM, Sperling RA, Johnson KA. Structural tract alterations predict downstream tau accumulation in amyloid-positive older individuals. Nat Neurosci. 2018 Mar;21(3):424-431. Epub 2018 Feb 5 PubMed.
Make a Comment
To make a comment you must login or register.