Toxic Synuclein Corrupts Native in Wild-Type Mice
Quick Links
The spread of toxic α-synuclein through the brain is not just for transgenic mice anymore. A new study from Virginia Lee and colleagues at the University of Pennsylvania School of Medicine, Philadelphia, suggests that toxic α-synuclein creeps between functionally connected areas in the wild-type mouse brain, killing dopaminergic neurons and causing motor difficulties in the process. As reported in the November 16 Science, one tiny injection of misfolded, fibrillar, synthetic α-synuclein recruits native protein and converts it to a pathogenic form, setting off a neurodegenerative cascade. Since the mouse expresses only wild-type α-synuclein of its own species, as do most humans with the disease, this injection model could be the closest yet to simulate pathogenic protein templating in sporadic forms of Parkinson’s disease. Results could have implications for treatments targeting protein transfer, write the authors.
“This study more or less nails down the causation of many idiopathic forms of Parkinson’s disease,” said Lary Walker, Emory University, Atlanta, Georgia, who was not involved in the work. “It shows that this process can be initiated simply by a misfolded and aggregated seed that induces further protein misfolding and aggregation.”
In recent years, evidence has grown to suggest that toxic amyloidogenic proteins, such as Aβ and tau, pass between neurons along functionally connected pathways, corrupting their normal counterparts as they go (see ARF related news story; ARF news story; de Calignon et al., 2012). Scientists thought the same might be true of α-synuclein. Researchers led by Seung-Jae Lee of Konkuk University in Seoul, South Korea, and Eliezer Masliah of the University of California, San Diego, reported cell-to-cell transmission of α-synuclein in neuronal cultures (see ARF related news story). Virginia Lee and colleagues found similar transmission in cells (see ARF related news story) and saw toxicity spread after injecting fibrils in transgenic mice that express mutant human α-synuclein (see ARF related news story). No one had yet shown that toxic synuclein could corrupt its normal counterparts in wild-type mice. That was the next logical step, she said. Just recently researchers at Rakez Kayed's lab showed that oligomers of human tau convert endogenous tau to toxic forms in mice (see Lasagna-Reeves et al., 2012).
First author Kelvin Luk and colleagues injected a small amount of misfolded, synthetic mouse α-synuclein into the dorsal striatum of wild-type C57BL6/C3H mice. After one month, hyperphosphorylated α-synuclein (an indication of synuclein-laden Lewy bodies) showed up around the injection site and in connected areas on the same side of the brain, including cortical layers IV and V and the olfactory bulb. As Lewy bodies in these areas multiplied over the next three to five months, inclusions also began turning up in additional connected areas—the neocortex, ventral striatum, thalamus, and occipital cortex. The pattern suggested that the injected fibrils acted as seeds that converted native α-synuclein to a pathogenic form, which then aggregated, crept through functionally linked areas, and converted more native protein. Pathology also spread the same way after injection of two other wild-type mice—CD1 and C57BL6/SJL. Researchers call this process templating (Hardy and Revesz, 2012).
The toxic synuclein further spread to dopaminergic neurons in the substantia nigra pars compacta, which is hit hard in Parkinson’s, causing the well-known motor deficits. Gradually, these neurons started dying and dopamine concentrations fell (see image below). After five months, injected mice performed worse than controls on rotarod and wire-hang tests of motor coordination, balance, and strength.
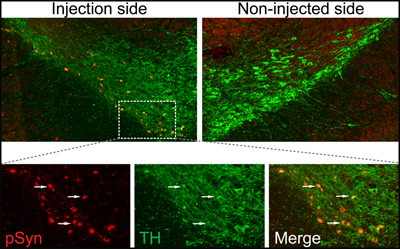
Templated Synuclein Misfolding
Dopaminergic mouse neurons display Lewy body-like aggregates (red) and reduced dopamine (green) on the same side of the brain as injected, misfolded α-synuclein. Image courtesy of Kelvin Luk, University of Pennsylvania Perelman School of Medicine
“The study demonstrates the direct link between the formation of Lewy bodies and the demise of dopaminergic cells,” said Lee. It also confirms that “spread of the pathology is not random; it follows areas that are functionally connected to each other.” She and her team are currently letting these mice age further to see if they begin to show cognitive deficits late in life, as do people with α-synucleinopathies. The injection model may also be suitable for testing therapies that target protein transmission in sporadic forms of the disease, said Lee.
Injection of the preformed α-synuclein fibrils could mimic a process that, in humans, occurs in the context of aging, said Walker. “As cells get old, they become less able to deal with protein misfolding.” He suggested that at some point, seeds of aggregated proteins accumulate and then move to other brain areas where they encounter normal proteins and convert those to toxic forms.
The next important step is to decipher the actual mechanism by which these misfolded proteins transfer between neurons, said Marc Diamond, Washington University, St. Louis, Missouri. “How do aggregates get into a neuron and how do they seed? How do they contact the normal protein? How and where does that conversion event happen in the cell, and how do aggregates get out?” Answers to these questions could help guide therapies that target pathological spread of proteins in PD, AD, and related neurodegenerative diseases, he told Alzforum.
The ability of wild-type α-synuclein to convert to a pathological form and then spread fits in with the idea that pathology actually starts in the gut, said Jeffrey Kordower, Rush University, Chicago, Illinois, who was not involved with this particular study. Developed by Heiko Braak and considered by some to be far fetched initially, this idea has gained support recently (see ARF related news story). The enteric nervous system is riddled with Lewy bodies long before they are found in the brain or before motor symptoms emerge (see Braak et al., 2006).—Gwyneth Dickey Zakaib
References
News Citations
- Mice Tell Tale of Tau Transmission, Alzheimer’s Progression
- Special Issue Explores Link Between Metabolic Disease and Dementia
- Research Brief: α-synuclein Spoils the Neural Neighborhood
- Modeling Sporadic PD in a Dish?
- Synthetic Synuclein Corrupts Native Along Mouse Brain Networks
- Parkinson’s: Thinking Outside the Brain’s Black Box
Paper Citations
- de Calignon A, Polydoro M, Suárez-Calvet M, William C, Adamowicz DH, Kopeikina KJ, Pitstick R, Sahara N, Ashe KH, Carlson GA, Spires-Jones TL, Hyman BT. Propagation of tau pathology in a model of early Alzheimer's disease. Neuron. 2012 Feb 23;73(4):685-97. PubMed.
- Lasagna-Reeves CA, Castillo-Carranza DL, Sengupta U, Guerrero-Munoz MJ, Kiritoshi T, Neugebauer V, Jackson GR, Kayed R. Alzheimer brain-derived tau oligomers propagate pathology from endogenous tau. Sci Rep. 2012;2:700. PubMed.
- Hardy J, Revesz T. The spread of neurodegenerative disease. N Engl J Med. 2012 May 31;366(22):2126-8. PubMed.
- Braak H, De Vos RA, Bohl J, Del Tredici K. Gastric alpha-synuclein immunoreactive inclusions in Meissner's and Auerbach's plexuses in cases staged for Parkinson's disease-related brain pathology. Neurosci Lett. 2006 Mar 20;396(1):67-72. PubMed.
Other Citations
Further Reading
Papers
- Goedert M, Clavaguera F, Tolnay M. The propagation of prion-like protein inclusions in neurodegenerative diseases. Trends Neurosci. 2010 Jul;33(7):317-25. PubMed.
- Hansen C, Li JY. Beyond α-synuclein transfer: pathology propagation in Parkinson's disease. Trends Mol Med. 2012 May;18(5):248-55. PubMed.
- Olanow CW, Prusiner SB. Is Parkinson's disease a prion disorder?. Proc Natl Acad Sci U S A. 2009 Aug 4;106(31):12571-2. PubMed.
- Eisenberg D, Jucker M. The amyloid state of proteins in human diseases. Cell. 2012 Mar 16;148(6):1188-203. PubMed.
News
- Special Issue Explores Link Between Metabolic Disease and Dementia
- Research Brief: α-synuclein Spoils the Neural Neighborhood
- Modeling Sporadic PD in a Dish?
- Synthetic Synuclein Corrupts Native Along Mouse Brain Networks
- Aβ Sufficient for Seeding—But Is It a Prion?
- Proteopathic Seeds and Neurodegenerative Diseases
- Mice Tell Tale of Tau Transmission, Alzheimer’s Progression
- Parkinson’s: Thinking Outside the Brain’s Black Box
- Parkinson's: It Started With a Gut Feeling
- An α-Synuclein Twist—Native Protein a Helical Tetramer
Primary Papers
- Luk KC, Kehm V, Carroll J, Zhang B, O'Brien P, Trojanowski JQ, Lee VM. Pathological α-synuclein transmission initiates Parkinson-like neurodegeneration in nontransgenic mice. Science. 2012 Nov 16;338(6109):949-53. PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
This is a very important paper that further supports the role of a prion-like propagation of proteins involved in neurodegenerative disorders. This is the first study that has been able to show propagation of α-synuclein after a single injection in non-transgenic wild-type animals; this is rather remarkable.
Previous studies have shown propagation, but in α-synuclein transgenic mice. Others have tried to propagate synuclein in non-transgenic mice but encountered difficulties. Thus, the question arises as to what is unique in this study by the group of Virginia Lee. One possibility is the characteristics of the α-synuclein seeds they developed. They might represent a strain more prone to propagate than others. Such seed strain properties have been shown for the prion protein.
Hertie Institute for Clinical Brain Research, University of Tübingen, and DZNE Tübingen
These are very impressive results from Virginia Lee and colleagues, and build on the previous work of the same group. The fact that synthetic material can act as a seed in wild-type mice opens a myriad of experimental approaches and avoids confounding factors such as overexpression of host proteins and the necessity of using brain-derived material as seed. This work will further advance the understanding of the propagation and spreading of protein misfolding diseases. It is becoming increasingly clear that protein aggregates associated with neurodegenerative disorders are found to have prion-like properties and, once misfolded, initiate a cascade of corruptive templating and related pathology: common mechanism, common amyloid principles (Eisenberg and Jucker, 2012).
References:
Eisenberg D, Jucker M. The amyloid state of proteins in human diseases. Cell. 2012 Mar 16;148(6):1188-203. PubMed.
Banner Sun Health Research Institute
This is an impressive result, agreed. Just would like to point out that the most likely and earliest entry point for an exogenous process would be through the olfactory bulb, not the gut. Our thorough survey of the peripheral nervous system in subjects with PD, DLB, ADLB, and ILBD, as well as aged normal controls, found no case where peripheral nervous system synucleinopathy was present in the absence of central nervous system synucleinopathy.
References:
Beach TG, Adler CH, Sue LI, Vedders L, Lue L, White Iii CL, Akiyama H, Caviness JN, Shill HA, Sabbagh MN, Walker DG, . Multi-organ distribution of phosphorylated alpha-synuclein histopathology in subjects with Lewy body disorders. Acta Neuropathol. 2010 Jun;119(6):689-702. PubMed.
Make a Comment
To make a comment you must login or register.