Grow that Axon: C9ORF72 Function Revealed?
Quick Links
While mutated C9ORF72 leads to some forms of amyotrophic lateral sclerosis (ALS) and frontotemporal degeneration (FTD), scientists still wonder what the healthy protein does. In the October 10 Nature Neuroscience online, scientists led by Michael Sendtner, University Hospital of Wuerzburg, Germany, suggest that it modulates actin dynamics. Motor neurons with sparse C9ORF72 generated fewer actin filaments and formed them very slowly, which truncated axons and hindered growth cones. Reduced C9ORF72 activity could contribute to the pathology of ALS and FTD, the authors conclude. The field has been debating whether loss of normal C9ORF72 function or the toxic peptides and RNAs made from a nucleotide expansion in the gene cause disease.
“This is one of the best studies I’ve seen so far on the native function of C9ORF72,” said Brian Freibaum, St. Jude Children’s Research Hospital, Memphis. “I think this [actin regulation] is a legitimate function of C9ORF72, and its decreased activity is probably causing further pathogenesis of the disease.”
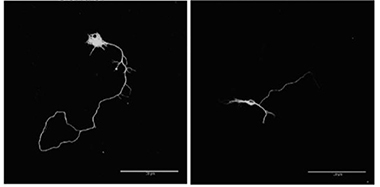
Stunted.
In mouse motor neurons (left), knocking down C9ORF72 truncates axons (right). [Courtesy of Sivadasan et al., Nature Neuroscience.]
C9ORF72 was first identified as a joint ALS and FTD gene in 2011 (Sep 2011 news). It carries a GGGGCC hexanucleotide repeat in intron 1 that expands in some people with these diseases. Healthy people carry fewer than 24 repeats, while patients carry hundreds or thousands. For the last five years, scientists have tried to figure out how this expansion leads to disease, focusing mainly on the extra-long mRNAs that huddle together into foci, or the dipeptide repeat proteins that are translated from the repeats and aggregate (Jan 2013 conference news; Oct 2013 news; Feb 2013 news). Few studies have examined the normal function of C9ORF72, even though patients with the repeat expansion end up with about half the normal amount of the protein. Could that cause problems that contribute to pathology?
To find out, first author Rajeeve Sivadasan and colleagues checked what would happen if they up- or downregulated C9ORF72 in mouse primary embryonic motor neurons. They focused on motor neurons because this cell type bears the brunt of pathology in ALS. When they overexpressed the protein, axons grew longer and had more robust growth cones. These guide the axon to its synaptic partners and spearhead regrowth after axon injury or disease. Reducing C9ORF72 with shRNA shortened axons and shrank growth cones (see image above).
The researchers wondered how C9ORF72 affected axon length. Using mass-spectrometry-based proteomics, they looked to proteins that interact with C9ORF72. Several regulators of actin dynamics emerged. Actin polymerizes into cytoskeletal filaments that give cells their shape and help them move and divide. Actin is essential for axon growth. To test whether loss of C9ORF72 impairs actin dynamics, Sivadasan used shRNAs to knock down C9ORF72 expression in cultured mouse motor neurons. Then they examined actin, which they had labeled with green florescent protein. Live imaging revealed that fewer filaments formed and actin extension in growth cones slowed to a crawl in the knockdowns. Filament growth also limped along in motor neurons derived from ALS patient iPSCs. “This is a really dramatic effect,” Sendtner told Alzforum. “Actin dynamics are severely disturbed in patient cells.”
How does C9ORF72 regulate actin? The researchers focused on a strong hit in their protein interactome, cofilin, which helps new actin chains form by clipping inert caps on the ends of filaments to expose actin for fresh polymerization. Cofilin also depolymerizes old filaments to release monomer that can be added to new actin chains. Cells turn off cofilin by phosphorylating it at serine 3. Was cofilin somehow hindered in the absence of C9ORF72?
Sivadasan found that knocking down C9ORF72 in mouse motor neurons enhanced cofilin phosphorylation. Immortalized lymphoblasts and iPSC-derived motor neurons from ALS patients with C9ORF72 expansions also contained more phosphorylated cofilin than normal. It was up threefold in postmortem patient brains compared with age-matched controls, as well. The researchers found that LIMK 1 and 2, kinases known to phosphorylate cofilin, were more active in mouse motor neurons that lacked C9ORF72 and in those derived from patient iPSCs.
The story quickly got even more complicated. These kinases are activated by Rac1, which itself is activated by Arf6. Both are GTPases regulated in turn by guanine nucleotide exchange factors (GEF). Previous structural homology studies predicted that C9ORF72 may belong to a large family of GEF and GEF-modulating proteins—the DENN family (Jan 2013 news). Could this be how C9ORF72 regulates actin?
Sivadasan found that C9ORF72 bound both Rac1 and Arf6, and that suppressing C9ORF72 led to more active, GTP-bound forms of both GTPases. With Arf6 in overdrive, it would phosphorylate cofilin, predicted the researchers. Indeed, when they knocked down Arf6 in iPSCs from patients, Rac1 activity fell. Expressing an inactive version of Arf6 in mouse motor neurons also rescued the effects of knocking down C9ORF72.
These results hint that reduced C9ORF72 function plays a role in ALS by hampering the actin dynamics crucial for cytoskeletal remodeling, synapse maintenance, and axon stability, wrote the authors. The finding jibes with other papers that have linked genetic variants in two actin-binding proteins, profilin 1 and filamin C, to ALS and FTD (July 2012 news; Nov 2014 news).
Sendtner thinks perturbing actin is but one way C9ORF72 expansions cause disease. “It remains to be seen which pathology is the ideal target for a therapy,” he said. He told Alzforum that the Arf6 pathway offers several points to interfere and reverse the block on actin-related processes. His group is now working to find the GEF that interacts with C9ORF72 to turn off Arf6.
“This is a very interesting finding about the role of the C9ORF72 protein in neuronal function,” write Robert Baloh, Cedars-Sinai Regenerative Medicine Institute, Los Angeles, to Alzforum. “It supports the growing idea that both loss- and gain-of-function mechanisms may be at play in ALS and FTD from C9ORF72 mutations.” The key next step is to see whether the actin effects act synergistically with RNA foci and dipeptide repeat proteins to promote neurodegeneration, he added.—Gwyneth Dickey Zakaib
References
News Citations
- Corrupt Code: DNA Repeats Are Common Cause for ALS and FTD
- Chicago—RNA Inclusions Offer Therapeutic Target in ALS
- RNA Deposits Confer Toxicity in C9ORF72 ALS
- RNA Twist: C9ORF72 Intron Expansion Makes Aggregating Protein
- C9ORF72 Function: Is the ALS Protein a Membrane Traffic Cop?
- Profilin Gene Is Actin’ in ALS
- Stream of Genetics Pushes FTD Research Forward
Further Reading
Papers
- Sullivan PM, Zhou X, Robins AM, Paushter DH, Kim D, Smolka MB, Hu F. The ALS/FTLD associated protein C9orf72 associates with SMCR8 and WDR41 to regulate the autophagy-lysosome pathway. Acta Neuropathol Commun. 2016 May 18;4(1):51. PubMed.
- Sudria-Lopez E, Koppers M, de Wit M, van der Meer C, Westeneng HJ, Zundel CA, Youssef SA, Harkema L, de Bruin A, Veldink JH, van den Berg LH, Pasterkamp RJ. Full ablation of C9orf72 in mice causes immune system-related pathology and neoplastic events but no motor neuron defects. Acta Neuropathol. 2016 Jul;132(1):145-7. Epub 2016 May 20 PubMed.
- DeJesus-Hernandez M, Mackenzie IR, Boeve BF, Boxer AL, Baker M, Rutherford NJ, Nicholson AM, Finch NA, Flynn H, Adamson J, Kouri N, Wojtas A, Sengdy P, Hsiung GY, Karydas A, Seeley WW, Josephs KA, Coppola G, Geschwind DH, Wszolek ZK, Feldman H, Knopman DS, Petersen RC, Miller BL, Dickson DW, Boylan KB, Graff-Radford NR, Rademakers R. Expanded GGGGCC hexanucleotide repeat in noncoding region of C9ORF72 causes chromosome 9p-linked FTD and ALS. Neuron. 2011 Oct 20;72(2):245-56. Epub 2011 Sep 21 PubMed.
- Gijselinck I, Van Langenhove T, van der Zee J, Sleegers K, Philtjens S, Kleinberger G, Janssens J, Bettens K, Van Cauwenberghe C, Pereson S, Engelborghs S, Sieben A, De Jonghe P, Vandenberghe R, Santens P, De Bleecker J, Maes G, Bäumer V, Dillen L, Joris G, Cuijt I, Corsmit E, Elinck E, Van Dongen J, Vermeulen S, Van den Broeck M, Vaerenberg C, Mattheijssens M, Peeters K, Robberecht W, Cras P, Martin JJ, De Deyn PP, Cruts M, Van Broeckhoven C. A C9orf72 promoter repeat expansion in a Flanders-Belgian cohort with disorders of the frontotemporal lobar degeneration-amyotrophic lateral sclerosis spectrum: a gene identification study. Lancet Neurol. 2012 Jan;11(1):54-65. PubMed.
News
- C9ORF72 Function: Is the ALS Protein a Membrane Traffic Cop?
- Methylation a Turn Off for Disease Gene C9ORF72?
- Tackling Mutant C9ORF72 Transcripts at the Source
- Paper Alert: Autoimmunity in Another C9ORF72-Deficient Mouse Strain
- New C9ORF72 Mice Develop Symptoms Resembling ALS/FTD
- C9ORF72 Dipeptide Repeat Not Enough for Motor Neuron Disease
Primary Papers
- Sivadasan R, Hornburg D, Drepper C, Frank N, Jablonka S, Hansel A, Lojewski X, Sterneckert J, Hermann A, Shaw PJ, Ince PG, Mann M, Meissner F, Sendtner M. C9ORF72 interaction with cofilin modulates actin dynamics in motor neurons. Nat Neurosci. 2016 Dec;19(12):1610-1618. Epub 2016 Oct 10 PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
No Available Comments
Make a Comment
To make a comment you must login or register.