Absent Aβ, Blood-Brain Barrier Breakdown Predicts Cognitive Impairment
Quick Links
Blood-brain barrier breakdown in the hippocampus might weaken cognition all on its own. According to a paper in the January 14 Nature Medicine, people with a damaged barrier were more likely to have early signs of cognitive impairment, regardless of any Aβ or tau pathology, or even other signs of vascular disease. Led by Berislav Zlokovic at the University of Southern California in Los Angeles, the study employed both fluid and imaging measures to gauge BBB damage, and cast a weakened barrier as an early biomarker for cognitive dysfunction.
- Researchers used both fluid and imaging measures to gauge integrity of blood-brain barrier.
- BBB damage correlated with cognitive impairment, independently of Aβ, tau, or vascular risk factors.
- Compromised barrier occurred in and around the hippocampus.
“This is an important study that identifies independent indices of BBB disruption as early markers of AD in a carefully studied patient cohort,” commented Costantino Iadecola of Weill Cornell Medical College in New York. “There is an urgent need to identify early biomarkers of disease and novel pathogenic factors, and this study represents a step forward in this direction.”
Vascular dysfunction in the brain is a well-recognized contributor to dementia. Its extent in the brain can range from the widespread vessel damage that underlies vascular dementia to subtler issues such as localized blood brain barrier leaks (Iadecola 2017; Sweeney et al., 2018). Animal studies have indicated that Aβ and tau pathology can inflict damage to the blood-brain barrier. In people, imaging studies have spotted erosion of the barrier in the early stages of AD (Montagne et al., 2017; Bennett et al., 2018; van de Haar et al., 2017; Feb 2015 webinar). However, researchers have yet to understand whether such early barrier breakdown is a cause or consequence of burgeoning AD pathology, or if it exacts a cognitive toll.
First author Daniel Nation and colleagues set out to disentangle the contributions of Aβ, tau, and BBB damage to cognitive decline. They studied two cohorts totaling 164 participants—one from USC and one from Washington University in St. Louis. Based on clinical dementia rating scores, 82 were cognitively normal with a CDR of zero, 65 were mildly impaired with CDR 0.5, and 17 with CDR 1 were deemed impaired. The researchers explicitly excluded participants with vascular dementia or vascular cognitive impairment, but included people who had other vascular risk factors, such as hypertension, diabetes, or cardiovascular disease. All of the volunteers underwent CSF sampling to assess Aβ and phospho-tau, and a subgroup of 35 also had Aβ-PET scans.
The researchers took a two-pronged approach to gauge BBB breakdown. First, they measured the CSF concentration of soluble platelet-derived growth factor receptor-β. This PDGFR-β fragment sheds from the surface of pericytes that line the blood-brain barrier when they respond to cell stress or injury. Zlokovic previously reported that CSF sPDGFR-β correlates with BBB damage (Sagare et al., 2015; Montagne et al., 2015). Second, in a subgroup of 73 volunteers, the researchers used dynamic contrast-enhanced (DCE) MRI to gauge BBB leakiness by tracking levels of an intravenously injected contrast agent.
Nation found sPDGFR-β levels increased with higher CDR scores. Cognitively normal people had significantly lower concentrations of the shed pericyte receptor than did people who were CDR 0.5. People with CDR 1 had higher concentrations still. The same relationship emerged between CDR scores and DCE-MRI: those with CDR 0.5 had higher levels of the contrast agent in the hippocampus and parahippocampal gyrus, but not in cortical or subcortical regions. The participants also took neuropsychological tests to assess cognitive domains including memory, attention/executive function, and language. People who scored poorly on at least one of 10 domains had higher measures of BBB damage than those who scored normally on all domains.
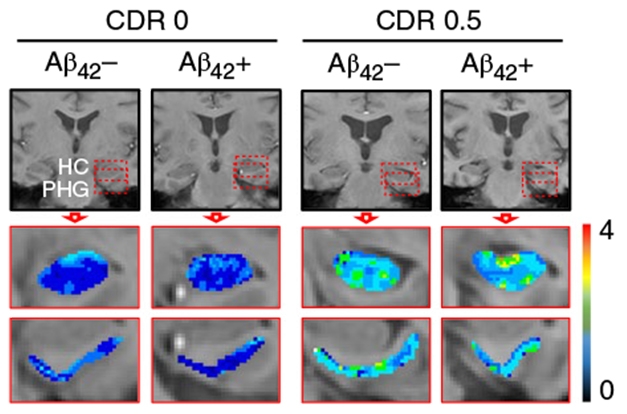
Leaks Strain Cognition. Compared with CDR zero, people with CDR 0.5 had a leakier blood-brain barrier in the hippocampus and parahippocampal gyrus, regardless of Aβ status. [Courtesy of Nation et al., Nature Medicine, 2019.]
Notably, these relationships between BBB damage and cognition held regardless of CSF Aβ or p-tau biomarker status. In fact, BBB damage predicted impairment regardless of hippocampal volume, vascular risk scores, or age as well. Interestingly, neither CSF sPDGFR-β nor DCE-MRI measures of BBB permeability changed with age. In all, the findings suggest that hippocampal BBB damage, and its cognitive toll, occurs outside of the normal trajectory of aging, and is distinct from impairment caused by overall vascular dysfunction, or Aβ and tau pathology, the researchers concluded.
Zlokovic told Alzforum he was surprised that BBB leakiness caused cognitive impairment independently of both AD pathology and vascular risk factors. The findings suggest that the hippocampus and surrounding regions are particularly vulnerable to barrier damage, just as they appear to be prone to tau pathology, he said. The molecular pathways that drive that vulnerability need further study, he added.
Kejal Kantarci of the Mayo Clinic in Rochester, Minnesota, noted the distinct vulnerability of hippocampal regions to barrier dysfunction. “As an independent factor, it is important to know why the breakdown of the BBB influences cognitive function mostly in the medial temporal lobe, but not in other brain regions that were studied,” she wrote. “Confirmation of the findings in cases with vascular cognitive impairment or vascular dementia is needed, which may demonstrate involvement of regions outside of the medial temporal lobe.”
Henrik Zetterberg of the University of Gothenburg in Sweden and Jonathan Schott of the Dementia Research Centre in London commented on the striking independence of hippocampal BBB damage from other vascular problems. This suggests that a predisposition to blood-brain barrier damage may be an independent risk factor for cognitive decline, they added. Zetterberg and Schott will jointly publish an editorial on the paper in an upcoming issue of the journal.
Iadecola wondered whether vascular problems in midlife could set the stage for a compromised barrier years down the line. “Midlife vascular risk factor are linked to late-life dementia, and processes that weaken the barrier could have conceivably started at that time, when the pathogenic processes driving AD also start,” he said.
What underlying syndrome explains the barrier leakage and cognitive impairment in people who tested negative for AD biomarkers? Only time will tell, Zlokovic said. He speculated that many could eventually become AD-biomarker-positive. He said the likelihood that these participants are on a trajectory toward vascular dementia is less likely, because that syndrome is characterized by distinct, and more widespread, vascular abnormalities than observed in this cohort.
Zlokovic proposed that CSF sPDGFR-β could serve as a useful biomarker for cognitive decline and neurodegenerative disease. His group is developing the CSF PDGFR-β assay to increase both sensitivity and standardization, he said.—Jessica Shugart
References
Webinar Citations
Paper Citations
- Iadecola C. The Neurovascular Unit Coming of Age: A Journey through Neurovascular Coupling in Health and Disease. Neuron. 2017 Sep 27;96(1):17-42. PubMed.
- Sweeney MD, Sagare AP, Zlokovic BV. Blood-brain barrier breakdown in Alzheimer disease and other neurodegenerative disorders. Nat Rev Neurol. 2018 Mar;14(3):133-150. Epub 2018 Jan 29 PubMed.
- Montagne A, Zhao Z, Zlokovic BV. Alzheimer's disease: A matter of blood-brain barrier dysfunction?. J Exp Med. 2017 Nov 6;214(11):3151-3169. Epub 2017 Oct 23 PubMed.
- Bennett RE, Robbins AB, Hu M, Cao X, Betensky RA, Clark T, Das S, Hyman BT. Tau induces blood vessel abnormalities and angiogenesis-related gene expression in P301L transgenic mice and human Alzheimer's disease. Proc Natl Acad Sci U S A. 2018 Feb 6;115(6):E1289-E1298. Epub 2018 Jan 22 PubMed.
- van de Haar HJ, Burgmans S, Jansen JF, van Osch MJ, van Buchem MA, Muller M, Hofman PA, Verhey FR, Backes WH. Blood-Brain Barrier Leakage in Patients with Early Alzheimer Disease. Radiology. 2017 Feb;282(2):615. PubMed.
- Sagare AP, Sweeney MD, Makshanoff J, Zlokovic BV. Shedding of soluble platelet-derived growth factor receptor-β from human brain pericytes. Neurosci Lett. 2015 Oct 21;607:97-101. Epub 2015 Sep 25 PubMed.
- Montagne A, Barnes SR, Sweeney MD, Halliday MR, Sagare AP, Zhao Z, Toga AW, Jacobs RE, Liu CY, Amezcua L, Harrington MG, Chui HC, Law M, Zlokovic BV. Blood-brain barrier breakdown in the aging human hippocampus. Neuron. 2015 Jan 21;85(2):296-302. PubMed.
Further Reading
Papers
- Sweeney MD, Zhao Z, Montagne A, Nelson AR, Zlokovic BV. Blood-Brain Barrier: From Physiology to Disease and Back. Physiol Rev. 2019 Jan 1;99(1):21-78. PubMed.
Primary Papers
- Nation DA, Sweeney MD, Montagne A, Sagare AP, D'Orazio LM, Pachicano M, Sepehrband F, Nelson AR, Buennagel DP, Harrington MG, Benzinger TL, Fagan AM, Ringman JM, Schneider LS, Morris JC, Chui HC, Law M, Toga AW, Zlokovic BV. Blood-brain barrier breakdown is an early biomarker of human cognitive dysfunction. Nat Med. 2019 Feb;25(2):270-276. Epub 2019 Jan 14 PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
Weill College Medicine, New York
This is an important study that identifies independent indices of BBB disruption as early markers of AD in a carefully studied patient cohort. There is an urgent need to identify early biomarkers of disease and novel pathogenic factors, and this study represents a step forward in this direction.
Assessing BBB function in humans is difficult. The authors used state-of-the-art MRI methods that they have used extensively, validated and refined to examine brain regions as small as hippocampal subregions. These methods, as imperfect as they might be, are still the best we have, and the present study highlights well their utility as early biomarkers of disease, as well as their potential to identify early pathogenic factors.
Beside its role as biomarker (discussed above), BBB dysfunction could also be an early pathogenic factor in AD. How BBB dysfunction promotes AD remains to be elucidated, but evidence for the proinflammatory effects of fibrinogen entering the brain has been provided. Importantly, BBB dysfunction is a manifestation of a more general neurovascular dysfunction involving the endothelium and the rest of the neurovascular unit, which, in addition to regulating flow and BBB permeability, also play a vital role in supporting synaptic function through trophic factors and nitric oxide.
The evidence suggesting independence of the BBB dysfunction from the CSF status of amyloid and tau is also of interest. However, a role of parenchymal Aβ and tau oligomers, not accurately reflected by their CSF levels, cannot be excluded. If the BBB dysfunction is indeed independent of the traditional pathogenic factors of AD, what might be its cause? Aging does not seem to correlate with the dysfunction, however, it would be of interest to examine midlife vascular risk factors, such as hypertension, etc.
Mayo Clinic
The effects of Alzheimer’s pathology on brain vasculature is an important topic and this study demonstrates that vascular endothelial disruption and the breakdown of the BBB is influencing cognitive performance independent of Alzheimer’s-related pathology. The investigators studied the medial temporal lobe including the hippocampus and parahippocampal gyrus, both of which are critical structures for early Alzheimer’s related pathology, in particular neurofibrillary tangle tau pathology. As an independent factor, it is important to know why the breakdown of the BBB influences cognitive function mostly in the medial temporal lobe, but not in other brain regions that were studied. Confirmation of the findings in cases with vascular cognitive impairment or vascular dementia is needed, which may demonstrate involvement of regions outside of the medial temporal lobe.
Maastricht University Medical Center
The investigators have to be congratulated for making a new major step in understanding the role of cerebral vascular pathology in cognitive decline. In subjects with and without mild cognitive impairment (MCI) they showed once more increases in blood-brain barrier (BBB) permeability to gadolinium MRI contrast agent in the hippocampus and platelet-derived growth factor receptor-β in the cerebral spinal fluid (CSF). It is easy to envision that BBB disturbs the homeostasis of brain tissue and leads to an ill-conditioned environment and neuronal dysfunction and eventually cell loss. Extending on previous research, they demonstrated no associations between measures of BBB breakdown and Aβ or tau, which were also derived from the CSF. With this observation they claim brain microvascular damage as an early biological marker of and contributor to cognitive dysfunction, independent of classical molecular markers of Alzheimer’s disease (AD). The study is very timely and adds to the increasing recognition that the vascular condition matters for brain function, also when no severe vascular lesions are apparent. Despite these advances in insight into the role of vascular pathology in cognitive impairment, there are a number of issues that need to be resolved and discussed.
The dynamic contrast-enhanced MRI (DCE-MRI) technique used to determine brain permeability is relatively new and complex, but yet applied for brain tissue by a number of clinical research groups worldwide. In normal healthy brain tissue the leakage of gadolinium contrast agent from the bloodstream into the brain tissue is negligible due to the blood-brain barrier. In case of BBB breakdown in normal-appearing brain tissue, the leakage will still be extremely slow and limited. The detection of this subtle leakage is very challenging and deals with noisy data. Currently, a number of independent research sites have demonstrated elevated BBB leakage of gadolinium contrast agents in patients with various forms of cognitive and/or cerebrovascular impairment and without overt brain lesions. However, the quantitative results vary strongly (more than one order of magnitude) among the different groups, which hampers objective comparisons between studies. Research sites diverge in the use of image acquisition and contrast agent protocols and image analysis methods. On the one hand standardization is needed, but on the other hand novel developments are greatly required to enhance the detection sensitivity of the ultra-low gadolinium levels in the parenchyma. In this context, it remains puzzling why the researchers find no leakage outside the medial temporal lobe, for instance in the white matter and neocortex, while others do. More global BBB breakdown would more directly fit to the decrements observed over the broader cognitive domains, other than episodic memory.
The current study focuses on a subpopulation of elderly people with the lowest cognitive or functional impairments (Clinical Dementia Rate 0.5) that can be objectified and those without noticeable impairments (CDR zero). A closer look at the subjects’ characteristics reveals that 50 percent of the participants (independent of CDR) suffer from hypertension. Though cardiovascular risk factors were well controlled for, and severe vascular injuries such as vascular cognitive impairment and vascular dementia were excluded, it is not unthinkable that these subjects have cerebral small-vessel-disease (cSVD), which is common in the elderly, especially those with hypertension. For such a population, inherent radiological features, including white-matter hyperintensities and microbleedings, are expected. White-matter intensities of cSVD often express in the deeper (periventricular) white matter, but not the hippocampus, and are known correlates of cognitive decline in aging and (preclinical) Alzheimer’s disease. Recent insights linked the white-matter hyperintensities to a malfunctioning neurovascular unit, where, among other matters, blood perfusion and permeability are tightly regulated. For the current study it remains to be shown to what extent the presence of white-matter hyperintensities affect or may explain the differences in the observed BBB disintegrity and cognitive impairment between the two groups.
A major achievement of the authors is investigating the correlation with CSF-derived protein markers of AD. Because in this cross-sectional study the variation in BBB leakage could not be explained by differences in Aβ or tau level, the influence of vascular pathology on cognitive impairment was considered independent from the key AD biomarkers Aβ and tau. Also, no correlation between hippocampal volume and cognitive impairment was found, emphasizing the early disease stage of cognitively impaired subjects. Taken together, this means that BBB breakdown and abnormal expressions of AD biomarkers, i.e., molecular fluid markers and neurodegeneration, are non-contemporary findings. Ideally, longitudinal studies are needed to conclude more firmly the timeline of the pathological cascade. Such studies may disentangle whether BBB breakdown truly initiates this cascade and its initial cognitive decrements, continues its extension, leads to vascular injuries, and/or adds to the full cognitive and functional disabilities of AD. This will be a long-lasting endeavour, also because only a limited number of subjects with MCI will converge into Alzheimer’s disease. Multiple cross-sectional studies on subjects in various disease stages ranging from mild cognitive impairment to full AD seem more feasible. Such studies, either longitudinal or cross-sectional, are, however, hampered by the quantification issues for DCE-MRI, as addressed above, which need further improvement and harmonization.
University of California, San Francisco
This paper provides important information on BBB functionality. However, as we continue discovering evidence of disruptions to brain homeostasis associated with the Alzheimer’s disease process, it is important to consider that the chronic, progressive neuroenergetic deficit, which evidence suggests may be primary to the process, has various downstream effects as the brain reprioritizes use of its diminished energy, with considerable energy being (mistakenly) diverted to inflammatory processes. There will be effects on the vast array of maintenance functions, including support for normal protein folding. Claudin-5 is a tight-junction protein that is key to the junctional adhesion molecules integral to the functionality of the blood-brain barrier. It will be important to understand the sequence of events wrought by the neuroenergetic deficit and factors that can influence the events in this process.
Make a Comment
To make a comment you must login or register.